Mannich Reaction
The reaction between aldimine (imines)containing aldehyde and α-methylene carbonyl yield β-amino carbonyl compound. This reaction is called mannich reaction or also called condensation reaction. An organic coupling reaction which is named on German chemist Carl Mannich is Mannich reaction.
“β-amino carbonyl compound is also called mannich base”
What is Mannich Reaction?
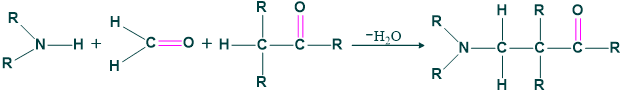
Primary and secondary aldimine give iminium ion.
Mannich Reaction Mechanism:
Step(01) formation of iminium ion:
In this step protonation of aldehydic group is done by acid and this protonated aldehyde is deprotonated by imine.
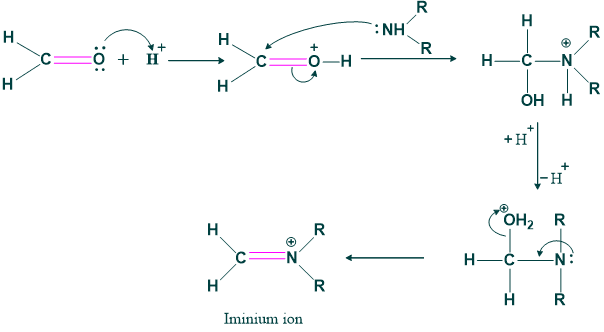
Step(02):
Here we form enol by keto-enol tautomerism .Here base OH not attack . Just tautomerism takes place.

Step(03):
The enol formed in the second step attacks on the iminium ion and forming the β-amino carbonyl compound or mannich base.
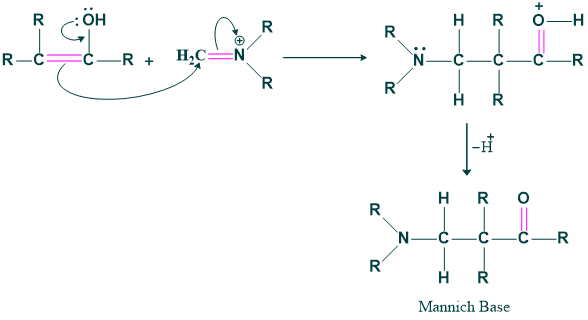
Application of Mannich Reaction:
Application of mannich reaction is as follows
- Synthesis of α,β-unsaturated compound
- Synthesis of alkaloids
- Robinson Schiff synthesis
Synthesis of α,β-unsaturated compound:
The mannich reaction of acetone with formaldehyde and di-methyliminium chloride in alcohol furnishes the salt of 3-(dimethylamino)-1-phenylpropane which can be easily eliminated to acrylophenone or 1-phenylprop-2-en-1-one an α,β-unsaturated carbonyl compound by converting it into quaternary salt and subsequent heating .This elimination is also called hoffmann elimination.
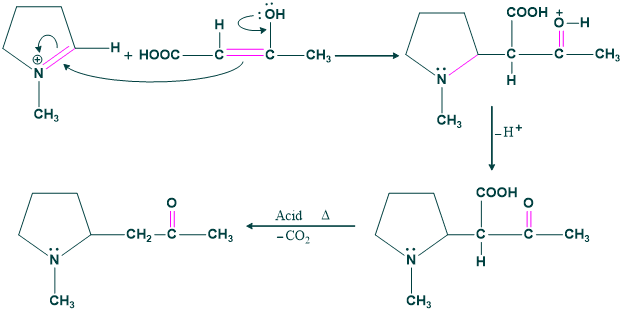
Synthesis of Alkaloids:
Alkaloids are natural products which are extracted from plants. They are basic in nature. That’s why they are called alkaloids .
Different alkaloids can be synthesized from different starting material via mannich reaction.
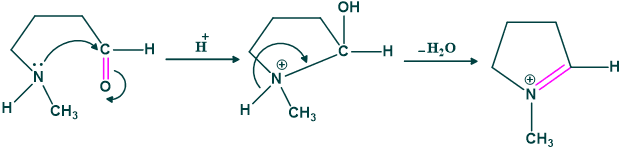
Step(01) hygrine formation:
Step(02):
Here tautomerization takes place.

Step(03):
Attack of enol on iminium ion.