Molecular Geometry & Molecular Shape:
What is Molecular Geometry?
Molecular geometry is the three-dimensional shape or arrangement of atoms in a molecule. Molecular geometry involves the positioning of the atoms surrounding the central atom as well as the bond angles between them.
OR
Molecular geometry is how a molecule appears in three dimensions.
Importance of Molecular Geometry In Chemistry
- Controls reactivity: Determines the reaction between molecules
- Decides polarity: Determines if the molecule is polar or nonpolar
- Influences properties: Determines boiling point, melting point, and solubility
- Important in biology: Shape decides how enzymes and drugs work
- Helps prediction: It is used to predict molecular behavior and stability
In short: Molecular geometry is important because it affects a molecule’s properties, reactions, and functions.
Molecular Shapes
When chemists talk about a molecule’s shape, they are referring to its geometry. Two molecules can share the same chemical formula yet behave completely differently because their atoms are arranged differently in 3D space.
The problem addressed by molecular geometry is, “What is the shape of a molecule?” And this shape determines most of its properties.
VSEPR Theory Fundamentals:
What is VSEPR?
The Valence Shell Electron Pair Repulsion (VSEPR) theory is a model employed in chemistry to determine the geometries of individual molecules through the determination of the number of valence electrons surrounding the central atom. The development of this model was mainly achieved by Ronald Gillespie and Ronald Nyholm in 1957. It built on earlier works by Nevil Sidgwick and Herbert Powell.
Core Principle:
“Electron pairs in the valence shell of a central atom repel one another and arrange themselves to be as far apart as possible, thereby minimizing electrostatic repulsion and determining the molecule’s three-dimensional shape.“
Electron pairs occupy space:
Both bonding pairs (shared between atoms) and lone pairs (non-bonding) occupy a region of space around the central atom and contribute to overall electron geometry.
Like Charges Repel:
All electron pairs are negatively charged, so they repel one another. The stable arrangement is the one that places pairs at maximum angular separation.
Lone Pairs Exert Greater Repulsion:
Lone pair electrons are attracted towards the nucleus more strongly and therefore lie in a larger solid angle than bonding electrons. The hierarchy of repulsions is: lone pair–lone pair > lone pair–bond pair > bond pair–bond pair.
Lone pair–lone pair > Lone pair–bonding pair > Bonding pair–bonding pair
Electron Pair Repulsion in Practice
Thinkabout water (H2O). Oxygen possesses four pairs ofelectrons in its outer shell – two pairs arebonded (to each hydrogen) and two areunshared. The perfect tetrahedral configurationpositions pairs 109.5° apart, but the two lone pairs reduce the H–O–H bond angle to around 104.5°. This compression, directly forecasted from the VSEPR repulsion hierarchy, correspondswith experimental data – a significantconfirmation of the theory outlined in Shriver & Atkins’ Inorganic Chemistry.
“The shape of a molecule is defined not just by the bonds, but by all electron pairs, includingboth bonding and non-bonding ones.”
Main Molecular Geometries (No Lone Pairs)
If there are no lone pairs present on the central atom, then the electron geometry and molecular geometry are the same.
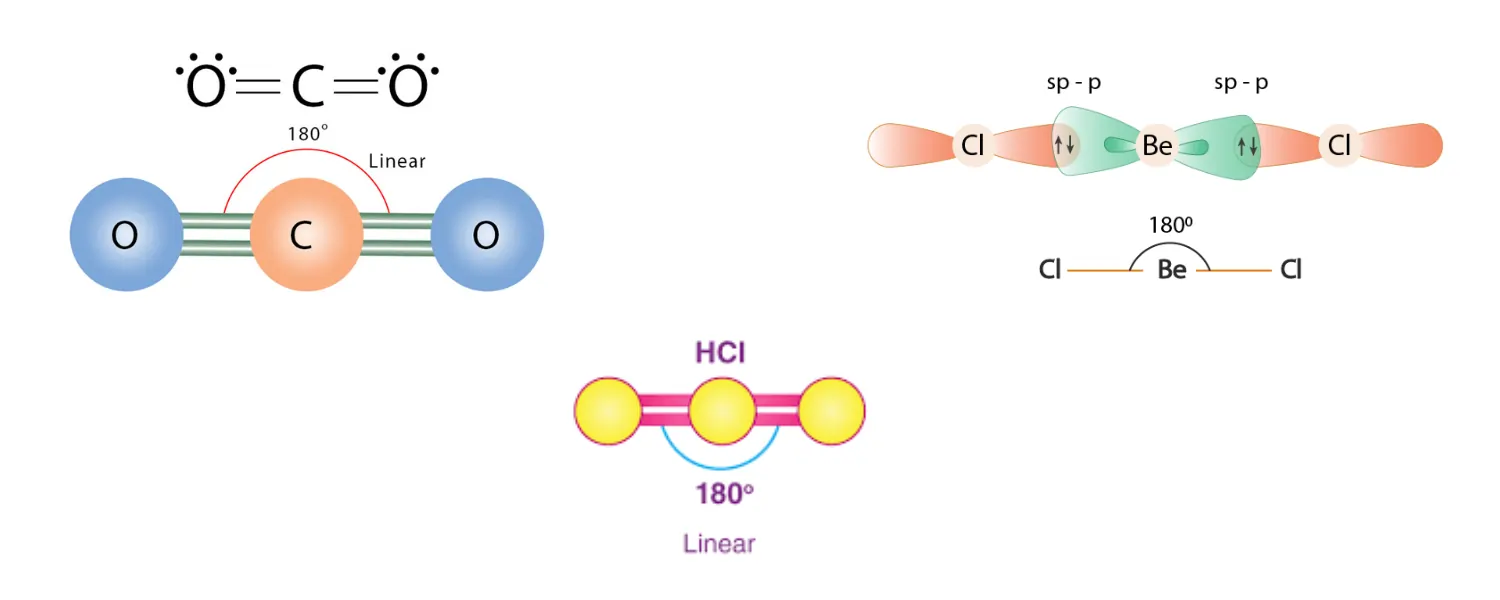
Linear – Bond Angle: 180°
Bonded atoms: 2 | Lone pairs on central atom: 0 | Example: CO₂, HCl, BeCl₂
The central atom is positioned midway between two bonding atoms along a straight axis. With just two regions of electrons present, maximum repulsion occurs when these regions point directly away from one another at angles of 180°.
Lesson in polarity for CO₂: Although carbon dioxide possesses two dipoles as a result of two polar C=O bonds, the perfect linearity results in both of these dipoles pointing in exactly opposite directions and canceling out each other. CO₂, therefore, is non-polar despite having polar bonds due to its symmetrical shap
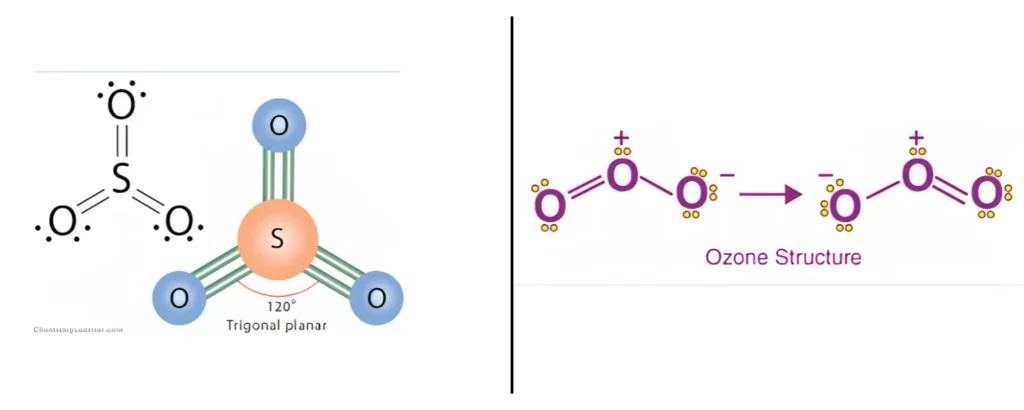
Trigonal Planar – Bond Angle: 120°
Bonded atoms: 3 | Lone pairs: 0 | Example: BF₃, SO₃, NO₃⁻
A trigonal planar molecular geometry model has one atom in the centre and three atoms at the corners of an equilateral triangle, known as peripheral atoms, all in the same plane. All three ligands in an ideal trigonal planar species are identical, and all bond angles are 120°. Molecules with three ligands which are non-identical deviate from this idealised geometry.
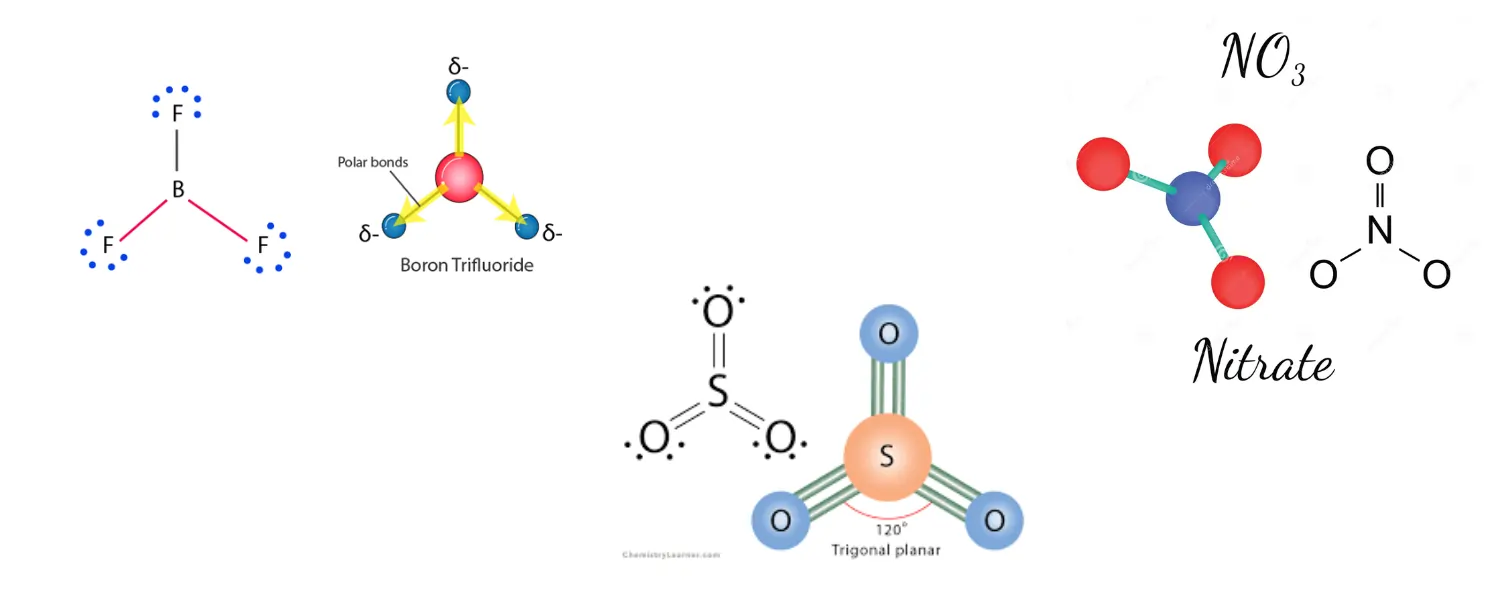
Boron’s exception: BF₃ is trigonal planar, but boron in this molecule has only six electrons around it — it is a well-known exception to the octet rule. This is stable because the empty p orbital on boron can accept electron density. Always note this when discussing BF
Tetrahedral — Bond Angle: 109.5°
Bonded atoms: 4 | Lone pairs: 0 | Example: CH₄, CCl₄, SiF₄
It is impossible for four electron domains to exist in one plane because geometrical constraints make it necessary that they occupy three dimensions.The perfect tetrahedral arrangement at 109.5° gives maximum separation.
Why tetrahedral geometry dominates chemistry and biology: Carbon forms four bonds and adopts tetrahedral geometry in virtually every organic molecule. The three-dimensional structure of proteins, carbohydrates, fats, and DNA all trace back to the tetrahedral carbon center. Without tetrahedral carbon, the molecular complexity necessary for life would be impossible
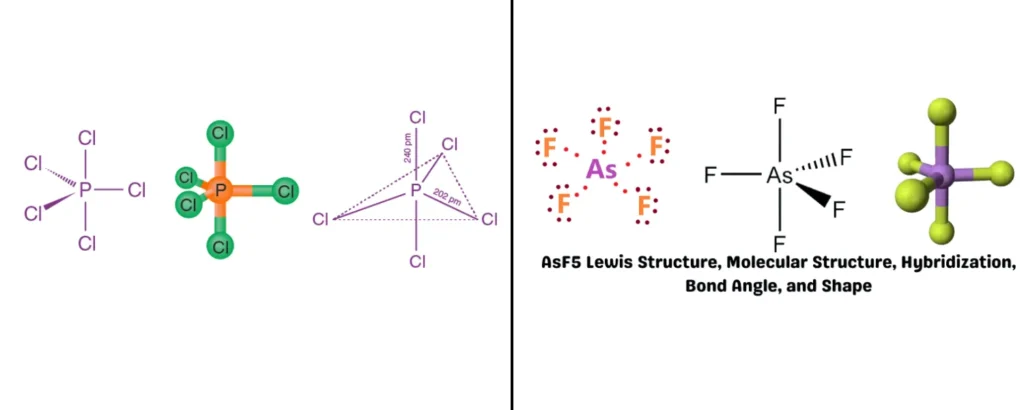
Trigonal Bipyramidal – Bond Angles: 120° (equatorial) and 90° (axial)
Bonded atoms: 5 | Lone pairs: 0 | Example: PCl₅, AsF₅
Three atoms reside in the triangular equatorial plane, which is separated by 120° between each other, while one more atom occupies both top and bottom (axial positions) at 90° relative to the atoms in the equatorial plane. Only in this type of geometry are not all bonding sites equal because the angles between atoms as well as the chemical environment are different for each position. Lone pair electrons that are present in the molecule prefer equatorial positions over axial.
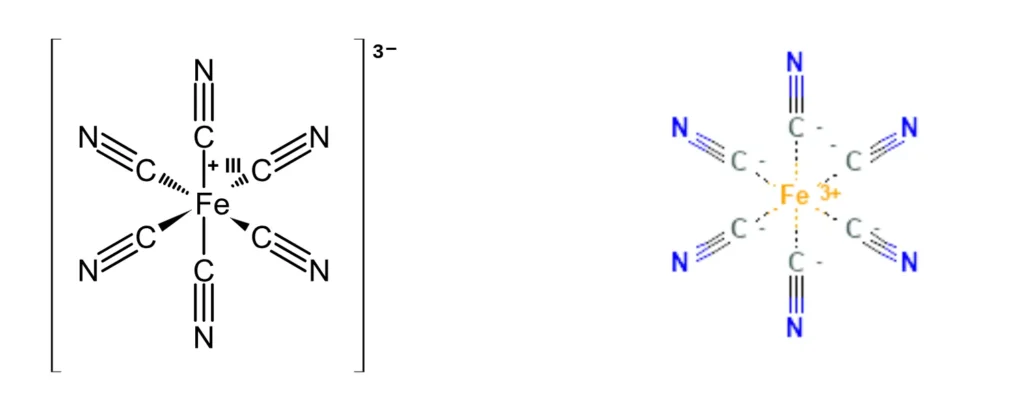
Octahedral — Bond Angle: 90°
Bonded atoms: 6 | Lone pairs: 0 | Example: SF₆, [Fe(CN)₆]³⁻
Six bonded atoms surround the central atom: four in a square plane, one directly above, one directly below. All bond angles are 90°. The shape resembles two square pyramids joined base-to-base.
Octahedral geometry is critical in transition metal coordination chemistry. Many industrial catalysts, biologically active metal centers, and colorful coordination complexes adopt octahedral arrangements.
Molecular Geometries with Lone Pairs:
Once lone pairs appear on the central atom, molecular geometry changes.
Bent — One Lone Pair (AX₂E₁)
Molecular geometry: Trigonal planar | Molecular shape: Bent | Bond angle: <120° (~117–119°) | Example: SO₂, O₃
Three electron domains arrange themselves in a trigonal planar electron geometry. One of those three positions is a lone pair — not a bonded atom. From the perspective of atomic positions only, the shape is bent. The lone pair pushes the two bonded atoms closer together, compressing the angle below 120°.
Trigonal Pyramidal (AX₃E₁)
Molecular geometry: Tetrahedral | Molecular shape: Trigonal pyramidal | Bond angle: ~107° | Example: NH₃, PCl₃, NF₃
Three electrons will take up positions in a trigonal planar electron arrangement. Out of these three positions, one will be occupied by a lone pair, which means that it will not be attached to any atoms. In terms of atomic position alone, the molecular geometry would be bent.
Trigonal Pyramidal (AX₃E₁)
Molecular geometry: Tetrahedral | Molecular shape: Trigonal pyramidal | Bond angle: ~107° | Example: NH₃, PCl₃, NF₃
The central atom has four electron regions. Three are bonds and one is a lone pair. The three bonded atoms form a pyramid shape.
Ammonia (NH₃) is the best example. The lone pair pushes the hydrogen atoms slightly closer together, reducing the bond angle from 109.5° to about 107°.
Important:
Trigonal planar → no lone pair, flat shape
Trigonal pyramidal → one lone pair, 3D shape
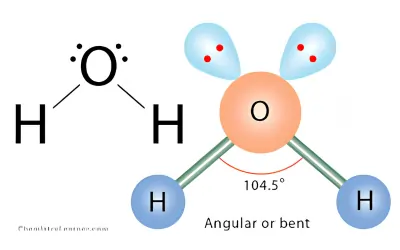
Bent — Two Lone Pairs (AX₂E₂)
Molecular geometry: Tetrahedral | Molecular shape: Bent | Bond angle: ~104.5° | Example: H₂O, H₂S
The central atom has four electron regions. Two are bonds and two are lone pairs. The lone pairs strongly repel the bonded atoms, making the bond angle even smaller.
Water (H₂O) is the most common example. Its bent shape makes water a polar molecule, which helps explain hydrogen bonding and many unique properties of water
The lone pair trend across four-domain molecules — a pattern directly tested in examinations:
Molecule | Lone Pairs | Bond Angle |
CH₄ (Tetrahedral) | 0 | 109.5° |
NH₃ (Trigonal pyramidal) | 1 | ~107° |
H₂O (Bent) | 2 | ~104.5° |
The AXE Method:
The AXE notation is a simple way to show how many atoms and lone pairs are attached to the central atom so you can predict the molecule’s shape.
A = the central atom
X = No. of bonded atoms attached to the central atom
E = No. of lone pairs on the central atom
Shape Of Molecules Chart – Complete AXE Reference Table
AXE | Electron Geometry | Molecular Shape | Bond Angle | Example |
AX₂ | Linear | Linear | 180° | CO₂ |
AX₃ | Trigonal planar | Trigonal planar | 120° | BF₃ |
AX₂E | Trigonal planar | Bent | <120° | SO₂ |
AX₄ | Tetrahedral | Tetrahedral | 109.5° | CH₄ |
AX₃E | Tetrahedral | Trigonal pyramidal | ~107° | NH₃ |
AX₂E₂ | Tetrahedral | Bent | ~104.5° | H₂O |
AX₅ | Trigonal bipyramidal | Trigonal bipyramidal | 90°, 120° | PCl₅ |
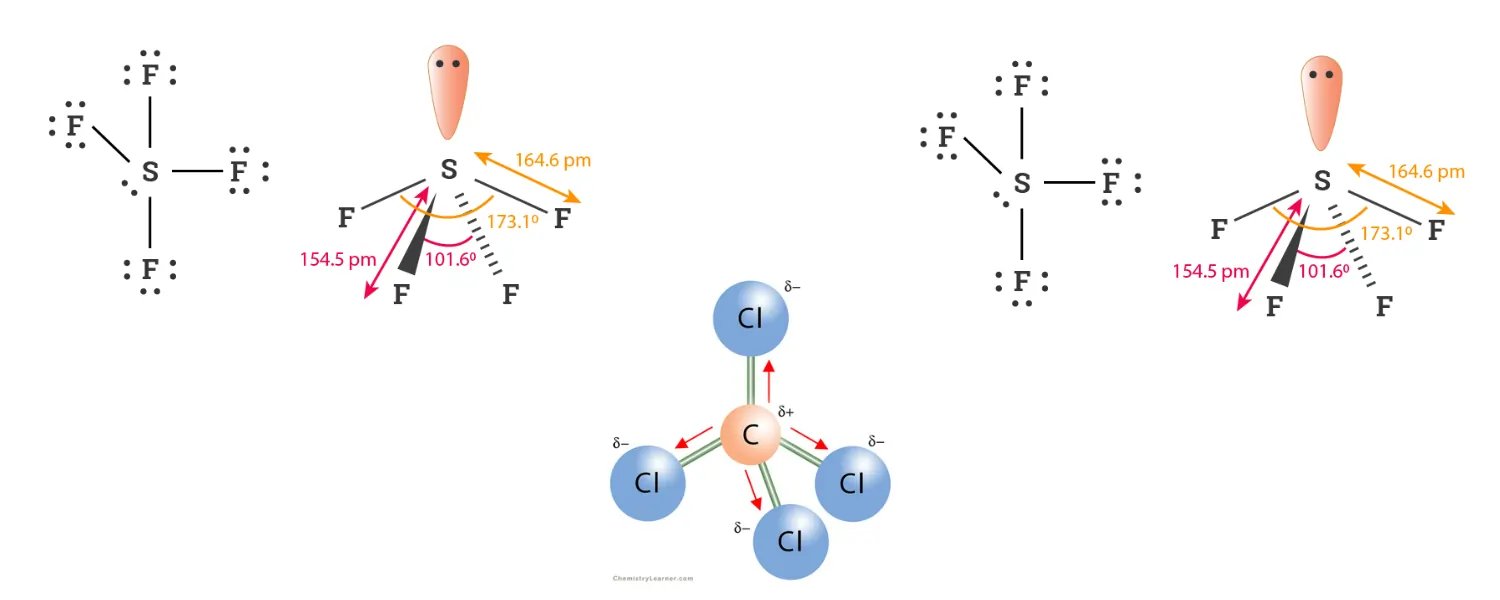
AX₄E | Trigonal bipyramidal | Seesaw | ~90°, ~120° | SF₄ |
AX₃E₂ | Trigonal bipyramidal | T-shaped | ~90° | ClF₃ |
AX₂E₃ | Trigonal bipyramidal | Linear | 180° | XeF₂ |
AX₆ | Octahedral | Octahedral | 90° | SF₆ |
AX₅E | Octahedral | Square pyramidal | ~90° | BrF₅ |
AX₄E₂ | Octahedral | Square planar | 90° | XeF₄ |
How to Determine Molecular Geometry: Step-by-Step
This simple four-step method can help you find the shape of almost any molecule.
Step 1: Draw the Lewis Structure
The Lewis structure shows how atoms and electrons are arranged in a molecule.
How to draw it:
- Choose the central atom
- Count total valence electrons
- Connect atoms with single bonds
- Add remaining electrons as lone pairs on outer atoms
- If needed, form double or triple bonds so atoms can complete their octet
- Place any leftover electrons on the central atom
Step 2: Count Electron Domains
An electron domain is any area of electrons around the central atom.Count them like this:
- Single bond = 1 domain
- Double bond = 1 domain
- Triple bond = 1 domain
- Lone pair = 1 domain
The total number of domains determines the basic arrangement around the central atom.
Domains | Arrangement | Ideal Bond Angle |
|---|---|---|
2 | Linear | 180° |
3 | Trigonal planar | 120° |
4 | Tetrahedral | 109.5° |
5 | Trigonal bipyramidal | 90°, 120° |
6 | Octahedral | 90° |
Step 3: Determine the Molecular Shape
Now focus only on the bonded atoms. Ignore lone pairs when naming the molecular shape.
Lone pairs still affect the shape because they push bonded atoms closer together.
Examples:
- CH₄ → tetrahedral
- NH₃ → trigonal pyramidal
- H₂O → bent
More lone pairs = smaller bond angles.
Step 4: Write the Final Answer
A complete answer usually includes:
- Molecular shape
- Bond angle
- Electron arrangement (optional but helpful)
Always remember:
- Electron arrangement includes all electron domains
- Molecular shape includes only bonded atoms
Bond Angles – Simple Explanation:
Bond angles are the angles between bonds around the central atom.
Factors That Affect Bond Angles
1. Electron Repulsion
Electron domains repel each other and spread out as much as possible to maintain a suitable distance to become stable.
2. Lone Pairs
Lone pairs repel more strongly than bonding pairs, so they reduce bond angles.
Example:
- CH₄ = 109.5°
- NH₃ = ~107°
- H₂O = ~104.5°
More lone pairs cause more compression.
3. Electronegativity
Highly electronegative atoms pull electrons toward themselves, which can slightly change bond angles.
Example:
- NH₃ has a bond angle of about 107°
- NF₃ has a smaller bond angle of about 102°
Common Bond Angles
Shape | Bond Angle | Example |
|---|---|---|
Linear | 180° | CO₂ |
Trigonal planar | 120° | BF₃ |
Tetrahedral | 109.5° | CH₄ |
Trigonal pyramidal | ~107° | NH₃ |
Bent | ~104.5° | H₂O |
Octahedral | 90° | SF₆ |
Worked Examples
These examples use the same four-step method to find molecular geometry and bond angles.
H2O Molecular Geometry
Step 1 – Draw the Lewis Structure
- Oxygen is the central atom
- Oxygen has 6 valence electrons
- Each hydrogen has 1 valence electron
- Total = 8 valence electrons
Oxygen forms two O–H bonds and keeps two lone pairs.
Step 2 — Count Electron Domains
- 2 bonding pairs
- 2 lone pairs
Total = 4 electron domains
Step 3 — Determine the Shape
4 electron domains give a tetrahedral arrangement.
Since there are 2 lone pairs:
- AX₂E₂
- Molecular shape = Bent
Step 4 — Determine Bond Angle
Two lone pairs compress the angle from 109.5° to about 104.5°.
Answer:
- Shape: Bent
- Bond angle: ~104.5°
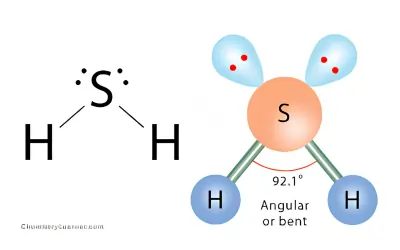
H2S Molecular Geometry
Step 1 – Draw the Lewis Structure
- Sulfur is the central atom
- Sulfur has 6 valence electrons
- Each hydrogen has 1 valence electron
- Total = 8 valence electrons
Sulfur forms two S–H bonds and keeps two lone pairs.
Step 2 – Count Electron Domains
Around the central sulfur atom:
- 2 bonding pairs
- 2 lone pairs
Total = 4 electron domainsains
Step 3 — Determine the Shape
4 electron domains give a tetrahedral arrangement.
Since there are 2 lone pairs:
- AX₂E₂
- Molecular shape = Bent
Step 4 — Determine Bond Angle
The two lone pairs compress the bond angle from the ideal tetrahedral angle (109.5°).
In H₂S, the bond angle is much smaller because sulfur is larger and bonding is weaker than in water.
Bond angle ≈ 92°
Answer:
- Shape: Bent
- Bond angle: ~92°
Why bond angle in h2s is less than h2o?
TheThe bond angle of H₂O (~104.5°) is greater than that of H₂S (~92°) because oxygen is more electronegative and smaller in size than sulfur. In water, the bonding electron pairs are drawn closer to the oxygen atom, resulting in stronger repulsion between electron pairs and a wider bond angle. In contrast, sulfur is larger and less electronegative, so the electron pair repulsions in H₂S are weaker, leading to a smaller bond angle.
BF3 Molecular Geometry
Step 1 – Draw the Lewis Structure
- Boron is the central atom
- Boron forms 3 single bonds with fluorine
- Boron has no lone pairs
Step 2 – Count Electron Domains
- 3 bonding pairs
- 0 lone pairs
Total = 3 electron domains
Step 3 – Determine the Shape
- AX₃
- Shape = Trigonal planar
Step 4 — Determine Bond Angle
No lone pairs means no compression.
Answer:
- Shape: Trigonal planar
- Bond angle: 120°
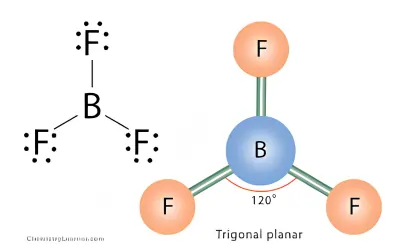
BF₃ is a flat molecule because all atoms lie in the same plane.
NH3 Molecular Geometry
Step 1 – Draw the Lewis Structure
- Nitrogen is the central atom
- Nitrogen forms 3 N–H bonds
- Nitrogen has 1 lone pair
Step 2 – Count Electron Domains
- 3 bonding pairs
- 1 lone pair
Total = 4 electron domains
Step 3 – Determine the Shape
- AX₃E
- Shape = Trigonal pyramidal
Step 4 – Determine Bond Angle
One lone pair reduces the angle from 109.5° to about 107°.
Answer:
- Shape: Trigonal pyramidal
- Bond angle: ~107°
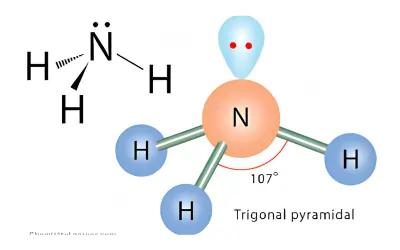
NH₃ looks like a pyramid with nitrogen at the top.
HCN Molecular Geometry
Step 1 – Draw the Lewis Structure
- Carbon is the central atom
- Hydrogen has 1 valence electron
- Carbon has 4 valence electrons
- Nitrogen has 5 valence electrons
- Total = 10 valence electrons
Carbon forms:
- One single bond with hydrogen (H–C)
- One triple bond with nitrogen (C≡N)
Nitrogen keeps one lone pair.
Step 2 – Count Electron Domains
Around the central carbon atom:
- 2 bonding regions
- One H–C bond
- One C≡N bond (counts as one domain)
- 0 lone pairs
Total = 2 electron domains
Step 3 – Determine the Shape
- 2 electron domains give a linear arrangement.
- Since there are no lone pairs on carbon:
- AX₂
- Molecular shape = Linear
Step 4 – Determine Bond Angle
Linear molecules have a bond angle of 180°.
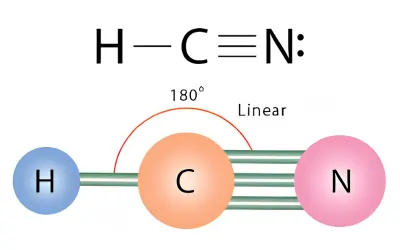
Answer:
- Shape: Linear
- Bond angle: 180°
CO2 Molecular Geometry
Step 1 – Draw the Lewis Structure
Carbon forms two double bonds with oxygen:
O=C=O
Carbon has no lone pairs.
Step 2 – Count Electron Domains
Each double bond counts as one domain.
- 2 bonding domains
- 0 lone pairs
Total = 2 electron domains
Step 3 – Determine the Shape
- AX₂
- Shape = Linear
Step 4 – Determine Bond Angle
No lone pairs means no compression.
Answer:
- Shape: Linear
- Bond angle: 180°
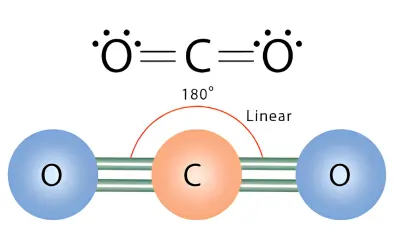
Although the C=O bonds are polar, CO₂ is non-polar because the molecule is perfectly symmetrical.
XeF4 Molecular Geometry
Step 1 – Draw the Lewis Structure
- Xenon is the central atom
- Xenon has 8 valence electrons
- Each fluorine has 7 valence electrons
- Total = 36 valence electrons
Xenon forms four single bonds with fluorine atoms and keeps two lone pairs.
Step 2 – Count Electron Domains
Around the central xenon atom:
- 4 bonding pairs
- 2 lone pairs
Total = 6 electron domainsains
Step 3 — Determine the Shape
6 electron domains give an octahedral arrangement.
Since there are 2 lone pairs:
- AX₄E₂
- Molecular shape = Square planar
Step 4 – Determine Bond Angle
Step 4 — Determine Bond Angle
The fluorine atoms are arranged in the same plane, giving bond angles of:
- Xe–F adjacent bond angle = 90°
Xe–F opposite bond angle = 180°
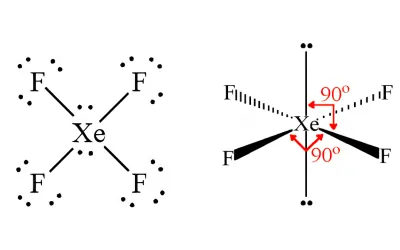
Answer:
- Shape: Square planar
- Bond angle: 90° and 180°
ClF3 Molecular Geometry
Step 1 – Draw the Lewis Structure
- Chlorine is the central atom
- Chlorine has 7 valence electrons
- Each fluorine has 7 valence electrons
- Total = 28 valence electrons
Chlorine forms three single bonds with fluorine atoms and keeps two lone pairs.
Step 2 – Count Electron Domains
Around the central chlorine atom:
- 3 bonding pairs
- 2 lone pairs
Total = 5 electron domainsainsains
Step 3 — Determine the Shape
5 electron domains give a trigonal bipyramidal arrangement.
Since there are 2 lone pairs:
- AX₃E₂
- Molecular shape = T-shaped
Step 4 — Determine Bond Angle
The lone pairs compress the bond angles slightly from the ideal 90° and 180°.
Bond angles are approximately:
Axial F – Cl – Equatorial F ≈ 90°
(slightly less than 90° because lone pairs compress the angle)°
Axial F – Cl – Axial F ≈ 180°
(slightly less due to lone pair repulsion)
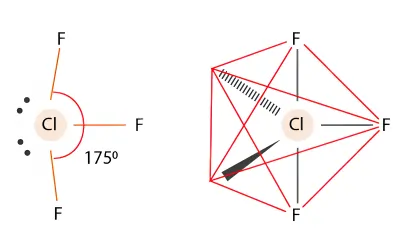
Answer:
- Shape: T-shaped
- Bond angle: approximately < 90° and < 180°
Molecular Geometry Chart:
Bonded (X) | Lone (E) | AXE | Electron Geometry | Molecular Shape | Bond Angle | Example |
2 | 0 | AX₂ | Linear | Linear | 180° | CO₂ |
3 | 0 | AX₃ | Trigonal planar | Trigonal planar | 120° | BF₃ |
2 | 1 | AX₂E | Trigonal planar | Bent | <120° | SO₂ |
4 | 0 | AX₄ | Tetrahedral | Tetrahedral | 109.5° | CH₄ |
3 | 1 | AX₃E | Tetrahedral | Trigonal pyramidal | ~107° | NH₃ |
2 | 2 | AX₂E₂ | Tetrahedral | Bent | ~104.5° | H₂O |
5 | 0 | AX₅ | Trigonal bipyramidal | Trigonal bipyramidal | 90°, 120° | PCl₅ |
4 | 1 | AX₄E | Trigonal bipyramidal | Seesaw | ~90°, ~120° | SF₄ |
3 | 2 | AX₃E₂ | Trigonal bipyramidal | T-shaped | ~90° | ClF₃ |
2 | 3 | AX₂E₃ | Trigonal bipyramidal | Linear | 180° | XeF₂ |
6 | 0 | AX₆ | Octahedral | Octahedral | 90° | SF₆ |
5 | 1 | AX₅E | Octahedral | Square pyramidal | ~90° | BrF₅ |
4 | 2 | AX₄E₂ | Octahedral | Square planar | 90° | XeF₄ |