Solubility Definition Chemistry
Solubility is the maximum amount of a substance (solute) that can be dissolved in a given amount of solvent at a specific temperature and pressure to make a homogeneous solution.
- It is usually expressed as grams of solute per 100 mL (or 100 g) of solvent at a given temperature.
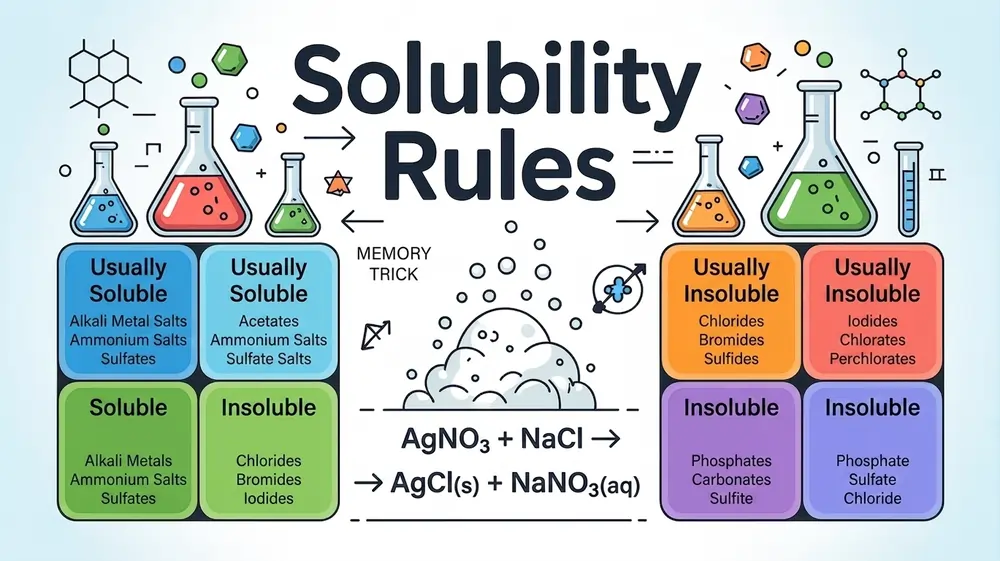
Solubility Rules Examples In Chemistry:
- Salt (NaCl) in water: About 36 g of table salt dissolves in 100 mL of water at 25°C. Beyond that, the salt stops dissolving and settles at the bottom.
- Sugar in tea: Sugar dissolves easily in hot water. More sugar can dissolve in hot tea than in cold tea, showing that solubility increases with temperature for most solids.
- Carbon dioxide (CO₂) in soda: CO₂ gas dissolves in water under high pressure to make carbonated drinks. When you open the bottle and pressure drops, the gas escapes – showing that gas solubility decreases with lower pressure.
- Oil in water: Oil does not dissolve in water at all. This makes oil an insoluble substance in water, since like dissolves like (polar dissolves polar; nonpolar dissolves nonpolar).
- Silver chloride (AgCl) in water: Only about 0.0002 g dissolves in 100 mL of water, making it classified as insoluble (or sparingly soluble) under solubility rules.
Why Solubility Rules Matter?
- Predicting Precipitation Reactions: Solubility rules help determine whether combining two solutions will result in a precipitate being formed. For instance, when AgNO₃ is combined with NaCl, an insoluble AgCl(s) compound forms. This can easily be predicted with solubility rules without performing any tests.
- Identifying Unknown Ions in a Solution: In the qualitative analysis process, solubility rules help determine the unknown ions in the mixture. This is achieved by using certain reagents, which react with certain ions to form insoluble products. Such ions include Cl⁻, SO₄²⁻, and Ba²⁺ among others.
- Essential for Exam Success (IGCSE, Edexcel, AP Chemistry): Solubility rules are a core topic in all major chemistry syllabuses. Students who memorize and apply them correctly can confidently answer precipitation, net ionic equation, and qualitative analysis questions in exams.
- Industrial and Real – World Applications: Solubility rules help regulate industrial operations like water purification (separation of ions through precipitation), metal extraction from minerals, and pharmaceutical manufacturing (ensuring the right level of drug solubility to facilitate appropriate drug absorption).
- Writing Correct Chemical Equations: Solubility rules are crucial when formulating the complete ionic equation and the net ionic equation. Without knowing which compounds are soluble or insoluble, it is impossible to correctly identify spectator ions or determine the driving force of a reaction.
Solubility Rules in Chemistry
SOLUBLE COMPOUNDS:
- Rule 1 – All Nitrates Are Soluble: All compounds containing the nitrate ion (NO₃⁻) are soluble in water without any exceptions. This means every metal combined with nitrate will dissolve completely, including calcium nitrate, lead nitrate, and silver nitrate.
- Rule 2 – All Ammonium Salts Are Soluble: There is absolutely no compound that contains the ammonium ion (NH₄⁺) that cannot be dissolved in water. Ammonium chloride, ammonium sulfate, ammonium carbonate – all dissolve completely regardless of the paired anion.
- Rule 3 – All Group 1 (Alkali Metal) Salts Are Soluble: All salts that containing lithium (Li⁺), sodium (Na⁺), potassium (K⁺), rubidium (Rb⁺), and cesium (Cs⁺) are soluble in water without any exceptions. This is one of the most reliable and important rules in all of chemistry.
- Rule 4 – All Chlorides Are Soluble (With Exceptions): The majority of compounds that have chloride ions (Cl⁻) are generally soluble in water. Soluble chlorides include sodium chloride, calcium chloride, iron chloride, and zinc chloride. There are few cases when these compounds are insoluble; for example, AgCl, PbCl₂, and Hg₂Cl₂ are insoluble.
- Rule 5 – All Bromides Are Soluble (With Exceptions): The majority of bromides are soluble in water. However, there is an exception, which is similar to chlorides where AgBr, PbBr₂, and Hg₂Br₂ are insoluble in water. All other bromides dissolve in water easily.
- Rule 6 – All Iodides Are Soluble (With Exceptions): Most iodides are soluble in water except for silver iodide (AgI), lead iodide (PbI₂), and mercury(1) iodide, which are insoluble. All other iodides are said to be soluble.
- Rule 7 – All Sulfates Are Soluble (With Exceptions): Compounds of the sulfate ion (SO₄²⁻) are generally soluble in water, except for BaSO₄, PbSO₄, and Hg₂SO₄. Sodium sulfate, copper sulfate, iron sulfate, and aluminum sulfate are some of the many examples of soluble sulfates. On the other hand, calcium sulfate (CaSO₄) and strontium sulfate (SrSO₄) are considered to be slightly soluble in water.
- Rule 8 – All Acetates Are Soluble: All compounds containing the acetate ion (CH₃COO⁻) are soluble in water with no exceptions. Copper acetate, lead acetate, and iron acetate are some examples that easily dissolve in water.
INSOLUBLE / SLIGHTLY SOLUBLE COMPOUNDS:
Rule 9 – Most Carbonates Are Insoluble: Most of the salts that contain the carbonate ion (CO₃²⁻) do not dissolve in water. These salts include calcium carbonate (CaCO₃), copper carbonate (CuCO₃), iron carbonate (FeCO₃), zinc carbonate (ZnCO₃), and lead carbonate (PbCO₃). Only Group 1 carbonates and those from ammonium ions are soluble in water. For instance, sodium carbonate (Na₂CO₃) and ammonium carbonate ((NH₄)₂CO₃.
Rule 10 – Most Hydroxides Are Insoluble: Most of the salts that have OH⁻ in their structure do not dissolve in water. Examples of such salts are iron hydroxide Fe(OH)₃, copper hydroxide Cu(OH)₂, zinc hydroxide Zn(OH)₂, and aluminum hydroxide Al(OH)₃. However, salts that contain OH⁻ but are Group 1 metals, for example, sodium hydroxide NaOH and potassium hydroxide KOH, completely dissolve in water. Barium hydroxide (Ba(OH)₂) and strontium hydroxide (Sr(OH)₂) are also soluble. Calcium hydroxide Ca(OH)₂ is a special case – it is only slightly soluble.
Rule 11 – Most Phosphates Are Insoluble: Almost all compounds containing the phosphate ion (PO₄³⁻) are insoluble in water. This includes calcium phosphate (Ca₃(PO₄)₂), iron phosphate (FePO₄), lead phosphate (Pb₃(PO₄)₂), and silver phosphate (Ag₃PO₄). Soluble phosphate salts can be found only for the elements of Group 1 (alkali metals) and ammonium, such as sodium phosphate (Na₃PO₄) and ammonium phosphate ((NH₄)₃PO₄).
Rule 12 – Most Sulfides Are Insoluble: Compounds having sulfide ion S²⁻ are generally insoluble in water. Some examples of sulfide salts that are insoluble in water include copper sulfide CuS, iron sulfide FeS, zinc sulfide ZnS, lead sulfide PbS, and silver sulfide Ag₂S. The exceptions are sulfides of Group 1 metals, Group 2 metals, and ammonium – for example sodium sulfide (Na₂S), calcium sulfide (CaS), and ammonium sulfide ((NH₄)₂S) are all soluble.
Rule 13 – Most Oxalates Are Insoluble: Most compounds containing the oxalate ion (C₂O₄²⁻) are insoluble in water. Calcium oxalate (CaC₂O₄) and silver oxalate (Ag₂C₂O₄) are common insoluble examples. The solubility of oxalates is limited to Group 1 metal ions and ammonium ions.
Rule 14 – Most Chromates Are Insoluble: Most of the compounds with the chromate ion (CrO₄²⁻) have low solubility or are insoluble. The examples of insoluble compounds include barium chromate (BaCrO₄), lead chromate (PbCrO₄), and silver chromate (Ag₂CrO₄). Sodium chromate and potassium chromate are exceptions and remain soluble.
Rule 15 – Silver Salts Are Mostly Insoluble: All silver (Ag⁺) salts, except for a very rare few, are insoluble in water. Silver chloride (AgCl), silver bromide (AgBr), silver iodide (AgI), silver carbonate (Ag₂CO₃), silver phosphate (Ag₃PO₄), and silver sulfide (Ag₂S) are examples of silver salts that are insoluble. However, the only commonly soluble silver salts are silver nitrate AgNO₃, silver acetate CH₃COOAg, and silver fluoride AgF.
QUICK MEMORY TIP For Solubility Rules
To remember which compounds are always soluble with no exceptions, remember this phrase:
“NASA K” – Nitrates, Ammonium, Sodium (and all Group 1), Acetates, and finally the “K” represents Potassium (also from Group 1).
They are always soluble without any exception and make up the core of all solubility guidelin.
Which Ions Are Soluble, Slightly Soluble, or Insoluble?
SOLUBLE IONS
1. Nitrate Ion (NO₃⁻): All nitrate compounds are soluble in water with absolutely no exceptions.There is no cation that can combine with the nitrate ion to form an insoluble substance.
Examples: Sodium nitrate (NaNO₃) – soluble
Potassium nitrate (KNO₃) – soluble
Calcium nitrate (Ca(NO₃)₂) – soluble
Silver nitrate (AgNO₃) – soluble
Lead nitrate (Pb(NO₃)₂) – soluble
2. Ammonium Ion (NH₄⁺): All ammonium salts dissolve in water without any exception. All types of anions combine with the ammonium ion and dissolve in the solvent.
Examples: Ammonium chloride (NH₄Cl) – soluble
Ammonium sulfate ((NH₄)₂SO₄) – soluble
Ammonium carbonate ((NH₄)₂CO₃) – soluble
Ammonium phosphate ((NH₄)₃PO₄) – soluble
3. Group 1 Alkali Metal Ions (Li⁺, Na⁺, K⁺, Rb⁺, Cs⁺): There are no exceptions to the fact that all Group 1 salts dissolve in water. It is one of the safest rules of solubility chemistry.
Examples: Lithium chloride (LiCl) – soluble
Sodium carbonate (Na₂CO₃) – soluble
Potassium hydroxide (KOH) – soluble
Sodium sulfide (Na₂S) – soluble
Potassium phosphate (K₃PO₄) – soluble
4. Acetate Ion (CH₃COO⁻): Every compound containing an acetate ion is soluble in water, regardless of which cation they are paired with.
Examples: Sodium acetate (CH₃COONa) – soluble
Copper acetate (Cu(CH₃COO)₂) – soluble
Lead acetate (Pb(CH₃COO)₂) – soluble
Iron acetate (Fe(CH₃COO)₃) – soluble
Ammonium acetate (CH₃COONH₄) – soluble
5. Chloride Ion (Cl⁻) – Mostly Soluble: Most chloride compounds are soluble in water.There are only three chlorides which are insoluble in nature while all others are soluble.
Soluble Examples: Sodium chloride (NaCl) – soluble
Calcium chloride (CaCl₂) – soluble
Iron(III) chloride (FeCl₃) – soluble
Zinc chloride (ZnCl₂) – soluble
Copper chloride (CuCl₂) – soluble
Insoluble Exceptions: Silver chloride (AgCl) – insoluble
Lead chloride (PbCl₂) – insoluble
Mercury(I) chloride (Hg₂Cl₂) – insoluble
6. Bromide Ion (Br⁻) – Mostly Soluble: Most bromides are soluble. Three bromides that cannot be dissolved are the exceptions that do not apply to most other bromides, just like what we see in chlorides.
Soluble Examples: Sodium bromide (NaBr) – soluble
Potassium bromide (KBr) – soluble
Calcium bromide (CaBr₂) – soluble
Magnesium bromide (MgBr₂) – soluble
Insoluble Exceptions: Silver bromide (AgBr) – insoluble
Lead bromide (PbBr₂) – insoluble
Mercry(I) bromide (Hg₂Br₂) – insoluble
7. Iodide Ion (I⁻) – Mostly Soluble: Most iodide compounds are soluble in water. The insoluble exceptions follow the same pattern as chlorides and bromides.
Soluble Examples: Sodium iodide (NaI) – soluble
Potassium iodide (KI) – soluble
Calcium iodide (CaI₂) – soluble
Zinc iodide (ZnI₂) – soluble
Insoluble Exceptions: Silver iodide (AgI) – insoluble
Lead iodide (PbI₂) – insoluble
Mercury(I) iodide (Hg₂I₂) – insoluble
8. Sulfate Ion (SO₄²⁻) – Mostly Soluble: Sulfate salts are generally soluble. A small group of sulfates are insoluble or slightly soluble.
Soluble Examples: Sodium sulfate (Na₂SO₄) – soluble
Copper sulfate (CuSO₄) – soluble
Aluminum sulfate (Al₂(SO₄)₃) – soluble
Iron sulfate (FeSO₄) – soluble
Zinc sulfate (ZnSO₄) – soluble
Insoluble Exceptions: Barium sulfate (BaSO₄) – insoluble
Lead sulfate (PbSO₄) – insoluble
Mercury(I) sulfate (Hg₂SO₄) – insoluble
Slightly Soluble: Calcium sulfate (CaSO₄) – slightly soluble
Strontium sulfate (SrSO₄) – slightly soluble
SLIGHTLY SOLUBLE IONS
9. Calcium Ion (Ca²⁺) – Mixed Solubility: Calcium salts exhibit dual properties as well because they may be soluble or insoluble/slightly soluble based on the ion present.
Soluble Examples: Calcium nitrate (Ca(NO₃)₂) – soluble
Calcium chloride (CaCl₂) – soluble
Slightly Soluble Examples: Calcium sulfate (CaSO₄) – slightly soluble
Calcium hydroxide (Ca(OH)₂) – slightly soluble
Insoluble Examples: Calcium carbonate (CaCO₃) – insoluble
Calcium phosphate (Ca₃(PO₄)₂) – insoluble
Calcium oxalate (CaC₂O₄) – insoluble
10. Fluoride Ion (F⁻) – Mixed Solubility: The behavior of fluoride salts in terms of solubility is different from other halides because of their variable nature.
Soluble Examples: Sodium fluoride (NaF) – soluble
Potassium fluoride (KF) – soluble
Silver fluoride (AgF) – soluble (unlike other silver halides)
Slightly Soluble / Insoluble Examples: Calcium fluoride (CaF₂) – slightly soluble
Magnesium fluoride (MgF₂) – slightly soluble
Barium fluoride (BaF₂) – slightly soluble
Lead fluoride (PbF₂) – slightly soluble
INSOLUBLE IONS
11. Carbonate Ion (CO₃²⁻) – Mostly Insoluble: Mostly all the compounds containing carbonates do not dissolve in water. The only exceptions are carbonates from Group 1 and ammonium carbonates which dissolve in water.
Solubility rules for carbonates : Calcium carbonate (CaCO₃) – insoluble
Copper carbonate (CuCO₃) – insoluble
Iron carbonate (FeCO₃) – insoluble
Zinc carbonate (ZnCO₃) – insoluble
Lead carbonate (PbCO₃) – insoluble
Silver carbonate (Ag₂CO₃) – insoluble
Soluble Exceptions: Sodium carbonate (Na₂CO₃) – soluble
Potassium carbonate (K₂CO₃) – soluble
Ammonium carbonate ((NH₄)₂CO₃) – soluble
12. Hydroxide Ion (OH⁻) – Mostly Insoluble: Hydroxide salts are usually insoluble. However, only a limited number of hydroxides are soluble in aqueous solutions.
Insoluble Examples: Iron(III) hydroxide (Fe(OH)₃) – insoluble
Copper hydroxide (Cu(OH)₂) – insoluble
Zinc hydroxide (Zn(OH)₂) – insoluble
Aluminum hydroxide (Al(OH)₃) – insoluble
Lead hydroxide (Pb(OH)₂) – insoluble
Magnesium hydroxide (Mg(OH)₂) – insoluble
Soluble Exceptions: Sodium hydroxide (NaOH) – soluble
Potassium hydroxide (KOH) – soluble
Barium hydroxide (Ba(OH)₂) – soluble
Lithium hydroxide (LiOH) – soluble
Slightly Soluble: Calcium hydroxide (Ca(OH)₂) – slighty soluble
13. Phosphate Ion (PO₄³⁻) – Mostly Insoluble: Most phosphate salts are not soluble in water. However, those of Group 1 and ammonium phosphates are soluble.
Insoluble Examples: Calcium phosphate (Ca₃(PO₄)₂) – insoluble
Zinc phosphate (Zn₃(PO₄)₂) – insoluble
Iron phosphate (FePO₄) – insoluble
Lead phosphate (Pb₃(PO₄)₂) – insoluble
Silver phosphate (Ag₃PO₄) – insoluble
Soluble Exceptions: Sodium phosphate (Na₃PO₄) – soluble
Potassium phosphate (K₃PO₄) – soluble
Ammonium phosphate ((NH₄)₃PO₄) – soluble
14. Sulfide Ion (S²⁻) – Mostly Insoluble: The majority of sulfide salts are insoluble in water. Those which are soluble in water include Group I, Group II, and ammonium sulfides.
Insoluble Examples: Copper sulfide (CuS) – insoluble
Iron sulfide (FeS) – insoluble
Zinc sulfide (ZnS) – insoluble
Lead sulfide (PbS) – insoluble
Silver sulfide (Ag₂S) – insoluble
Nickel sulfide (NiS) – insoluble
Soluble Exceptions: Sodium sulfide (Na₂S) – soluble
Potassium sulfide (K₂S) – soluble
Ammonium sulfide ((NH₄)₂S) – soluble
Calcium sulfide (CaS) – soluble
15. Chromate Ion (CrO₄²⁻) – Mostly Insoluble: The majority of the substances containing chromate ions are either insoluble or slightly soluble in water.
Insoluble Examples: Barium chromate (BaCrO₄) – insoluble
Lead chromate (PbCrO₄) – insoluble
Silver chromate (Ag₂CrO₄) – insoluble
Soluble Exceptions: Sodium chromate (Na₂CrO₄) – soluble
Potassium chromate (K₂CrO₄) – soluble
Ammonium chromate ((NH₄)₂CrO₄) – soluble
16. Oxalate Ion (C₂O₄²⁻) – Mostly Insoluble: The oxalate compounds are insoluble in water.
Insoluble Examples: Calcium oxalate (CaC₂O₄) – insoluble
Silver oxalate (Ag₂C₂O₄) – insoluble
Iron oxalate (FeC₂O₄) – insoluble
Lead oxalate (PbC₂O₄) – insoluble
Soluble Exceptions: Sodium oxalate (Na₂C₂O₄) – soluble
Potassium oxalate (K₂C₂O₄) – soluble
Ammonium oxalate ((NH₄)₂C₂O₄) – soluble
How to Use Solubility Rules: Step-by-Step?
WHAT YOU NEED BEFORE YOU START
Before applying solubility rules, you must have three things ready:
- It is essential to have a complete knowledge of what a cation (positively charged ion) and anion (negatively charged ion) are
- All of the solubility rules that have been memorized or that are readily accessible as a reference
- The ability to identify the ions present in any given ionic compound
Once you have these three things, applying solubility rules becomes a simple and systematic process every single time.
METHOD 1 – HOW TO PREDICT WHETHER A SINGLE COMPOUND DISSOLVES
Step 1 – Identify the Cation and Anion: Break the compound into its positive ion (cation) and negative ion (anion)
Example: Given compound – Calcium Carbonate (CaCO₃)
Cation = Ca²⁺ (calcium ion)
Anion = CO₃²⁻ (carbonate ion)
Step 2 – Check the Cation First: Consider the cation and ask the following questions in that order:
Question 1: Is it a Group 1 metal ion (Li⁺, Na⁺, K⁺, Rb⁺, Cs⁺)? If YES → the compound is soluble. Stop here. No need to check further.
Question 2: Is it an ammonium ion (NH₄⁺)? If YES → the compound is soluble. Stop here.
If neither of the above → move to Step 3 and check the anion.
Step 3 – Check the Anion:
Now look at the anion and apply the relevant solubility rule.
If the anion is nitrate (NO₃⁻): Compound is soluble – no exceptions.
If the anion is acetate (CH₃COO⁻): Compound is soluble – no exceptions.
If the anion is chloride (Cl⁻), bromide (Br⁻), or iodide (I⁻): Compound is soluble unless the cation is Ag⁺, Pb²⁺, or Hg₂²⁺ → then insoluble.
If the anion is sulfate (SO₄²⁻): Compound is soluble unless the cation is Ba²⁺, Pb²⁺, or Hg₂²⁺ → then insoluble. If cation is Ca²⁺ or Sr²⁺ → slightly soluble.
If the anion is carbonate (CO₃²⁻): Compound is insoluble unless cation is Group 1 or NH₄⁺ → then soluble.
If the anion is hydroxide (OH⁻): Compound is insolubleunless cation is Group 1, Ba²⁺, or Sr²⁺ → then soluble. If cation is Ca²⁺ → slightly soluble.
If the anion is phosphate (PO₄³⁻): Compound is insoluble unless cation is Group 1 or NH₄⁺ → then soluble.
If the anion is sulfide (S²⁻): Compound is insoluble unless cation is Group 1, Group 2, or NH₄⁺ → then soluble.
Step 4 – State Your Answer Clearly:
Always write your final answer as one of these three options:
Soluble – dissolves completely in water
Slightly soluble – dissolves only partially in water
Insoluble – does not dissolve in water (written as ↓ in equations)
Example : Is Barium Sulfate BaSO₄ soluble?
Step 1: Cation = Ba²⁺ / Anion = SO₄²⁻
Step 2: Ba²⁺ is not Group 1 and not NH₄⁺ → move to anion
Step 3: Anion is SO₄²⁻ → soluble unless cation is Ba²⁺, Pb²⁺, or Hg₂²⁺. Cation IS Ba²⁺.
Step 4: BaSO₄ is insoluble ↓
Example : Is Ammonium Phosphate (NH₄)₃PO₄ soluble?
Step 1: Cation = NH₄⁺ / Anion = PO₄³⁻
Step 2: NH₄⁺ → compound is soluble. Stop here.
Step 4: (NH₄)₃PO₄ is soluble
Example : Is Calcium Sulfate CaSO₄ soluble?
Step 1: Cation = Ca²⁺ / Anion = SO₄²⁻
Example : Is Calcium Sulfate CaSO₄ soluble?
Step 1: Cation = Ca²⁺ / Anion = SO₄²⁻
Step 2: Ca²⁺ is not Group 1 and not NH₄⁺ → move to anion
Step 3: Anion is SO₄²⁻ → slightly soluble when cation is Ca²⁺
Step 4: CaSO₄ is slightly soluble
THE ROLE OF SOLUBILITY RULES IN PRECIPITATION REACTIONS
Solubility rules are the key component without which it is impossible to make predictions regarding precipitation reactions in the field of chemistry. It is not possible to determine in advance whether the combination of two solutions will yield a precipitate or result in a clear solution without a reaction.
The connection between solubility rules and precipitation reactions works like this – when two ionic solutions are mixed, the ions recombine into new possible products. Solubility rules tell you instantly whether those new products are soluble (stay dissolved) or insoluble (form a precipitate). If at least one product is insoluble according to solubility rules, a precipitation reaction occurs.
TYPES OF EQUATIONS IN PRECIPITATION REACTIONS
Each and every precipitation reaction can be expressed in three different ways, depending on the degree of detail provided.
1. Molecular Equation
This is the easiest type. Here, complete chemical equations are written for both reactants and products without breaking them into ions. The state symbols are also used to indicate that a compound is in an aqueous or solid state.
Example: AgNO₃ (aq) + NaCl (aq) → AgCl (s) ↓ + NaNO₃ (aq)
2. Full Ionic Equation
All aqueous substances will be dissociated to the basic units or ions that make them up in this notation system. No solids are separated since they are not dissolved. All spectator ions are included in this equation.
Example: Ag⁺ (aq) + NO₃⁻ (aq) + Na⁺ (aq) + Cl⁻ (aq) → AgCl (s) ↓ + Na⁺ (aq) + NO₃⁻ (aq)
3. Net Ionic Equation
This is the most crucial and most instructive expression. This expression is generated by removing all the spectator ions from the complete ionic equation. The remaining expression contains only those ions which react to form the precipitate.
Example: Ag⁺ (aq) + Cl⁻ (aq) → AgCl (s) ↓
The net ionic equation is the same regardless of which soluble silver salt and which soluble chloride salt are used – because the actual chemical event is always just Ag⁺ meeting Cl⁻.
WHAT ARE SPECTATOR IONS?
Spectator ions are ions that are found in the solution before the reaction takes place but do not get involved in the reaction itself. These ions stay dissolved in water even after the reaction is over, and they look the same before and after the reaction, just like how they would be written in a complete ionic equation.
Example: In the reaction AgNO₃ (aq) + NaCl (aq) → AgCl ↓ + NaNO₃ (aq)
The spectator ions are Na⁺ and NO₃⁻ because they do not form the precipitate and remain dissolved throughout. The actual reaction is only between Ag⁺ and Cl⁻.
COLORS OF COMMON PRECIPITATES
Among the most interesting features associated with precipitation reactions is that many of the precipitates have their own unique coloration. It is very important to know what colors precipitates have as it plays a key role in lab work and tests.
White Precipitates: AgCl – silver chloride (white)
BaSO₄ – barium sulfate (white)
CaCO₃ – calcium carbonate (white)
PbSO₄ – lead sulfate (white)
Al(OH)₃ – aluminum hydroxide (white)
Mg(OH)₂ – magnesium hydroxide (white)
Zn(OH)₂ – zinc hydroxide (white)
BaCO₃ – barium carbonate (white)
CaSO₄ – calcium sulfate (white)
Yellow Precipitates: PbI₂ – lead iodide (bright yellow)
PbI₂ – lead iodide (bright yellow)
BaCrO₄ – barium chromate (yellow)
SnS – tin(II) sulfide (yellow-brown)
Blue Precipitates: Cu(OH)₂ – copper(II) hydroxide (pale blue)
Cu₃(PO₄)₂ – copper(II) phosphate (blue)
Green Precipitates: Fe(OH)₂ – iron(II) hydroxide (dirty green)
Ni(OH)₂ – nickel hydroxide (green)
MnS – manganese sulfide (green)
Cu₂Cl₂ – copper(I) chloride (pale green)
Brown / Rust Precipitates: Fe(OH)₃ – iron(III) hydroxide (rust brown)
MnO₂ – manganese dioxide (dark brown)
Black Precipitates: CuS – copper sulfide (black)
PbS – lead sulfide (black)
Ag₂S – silver sulfide (black)
FeS – iron sulfide (black)
NiS – nickel sulfide (black)
Cream / Pale Yellow Precipitates: AgBr – silver bromide (cream/pale yellow)
AgI – silver iodide (pale yellow)
MOST IMPORTANT PRECIPITATION REACTIONS WITH FULL WORKING:
Silver Nitrate + Sodium Chloride
Purpose: Test for chloride ions in solution
Molecular Equation: AgNO₃ (aq) + NaCl (aq) → AgCl ↓ (s) + NaNO₃ (aq)
Full Ionic Equation: Ag⁺ (aq) + NO₃⁻ (aq) + Na⁺ (aq) + Cl⁻ (aq) → AgCl (s) ↓ + Na⁺ (aq) + NO₃⁻ (aq)
Net Ionic Equation: Ag⁺ (aq) + Cl⁻ (aq) → AgCl (s) ↓
Precipitate: AgCl — white precipitate
Spectator Ions: Na⁺ and NO₃⁻
Solubility Rule Applied: Silver chloride is insoluble (Ag⁺ is an exception to the chloride solubility rule)
Observation: White precipitate forms immediately when solutions are mixed
Quantitative Solubility: Beyond the Basic Solubility Rules
INTRODUCTION
As far as the basic solubility rules mentioned above, we get a very straightforward answer, whether a particular compound is soluble, slightly soluble, or insoluble. But real-life situations require more accurate and quantitative answers. To what extent is a particular substance dissolved? What concentration will initiate precipitation? How will the presence of one ion influence the solubility of another?
That’s when the concept of quantitative solubility steps in. The essence of quantitative solubility lies not in the mere ability to say whether the solute is soluble, but rather in calculating the exact amount dissolved using certain formulas and equilibrium constants.
SOLUBILITY PRODUCT CONSTANT (Ksp)
What Is Ksp?
Ksp (solubility product constant) is an equilibrium constant which indicates the equilibrium existing between a sparingly soluble ionic compound and its dissociated ions in a saturated solution. It denotes the highest product value of concentrations of ions possible in a solution without the formation of a solid compound. Whenever a sparingly soluble ionic compound is put into water, it will eventually reach an equilibrium stage whereby the rate at which it is dissolving will be equal to the rate of precipitation.
For a general sparingly soluble salt:
MₐXᵦ (s) ⇌ aM^n⁺ (aq) + bX^m⁻ (aq)
The solubility product expression is:
Ksp = [M^n⁺]ᵃ × [X^m⁻]ᵇ
The concentration of the solid MₐXᵦ is not included in the expression because pure solids have constant activity equal to 1.
Why Is Ksp Important?
Ksp is important because it allows chemists to:
- Calculate the exact molar solubility of a compound in water
- Predict whether a precipitate will form when two solutions are mixed
- Determine how much of an ion remains in solution after precipitation
- Understand how factors like common ions and pH affect solubility
- Design separation procedures in analytical and industrial chemistry.
How to Write Ksp Expressions
Example : Silver Chloride (AgCl)
Dissolution: AgCl (s) ⇌ Ag⁺ (aq) + Cl⁻ (aq)
Ksp = [Ag⁺][Cl⁻]
Since one formula unit produces one Ag⁺ and one Cl⁻, both have exponent 1
Ksp Values for Common Ionic Compounds
A lower Ksp indicates lesser solubility, while a higher Ksp indicates greater solubility. There is a huge difference between them, ranging from compounds that are relatively soluble (Ksp = 10⁻⁴) to those that are extremely insoluble (Ksp = 10⁻⁵⁰).
Halides: AgF — 205 (very soluble – not truly sparingly soluble)
AgCl – 1.8 × 10⁻¹⁰ (insoluble)
AgBr – 5.0 × 10⁻¹³ (more insoluble than AgCl)
AgI – 8.5 × 10⁻¹⁷ (very insoluble)
PbCl₂ – 1.6 × 10⁻⁵ (slightly soluble)
PbBr₂ – 6.6 × 10⁻⁶ (slightly soluble)
PbI₂ – 9.8 × 10⁻⁹ (insoluble)
Sulfates: BaSO₄ – 1.1 × 10⁻¹⁰ (extremely insoluble)
PbSO₄ – 2.5 × 10⁻⁸ (insoluble)
CaSO₄ – 4.9 × 10⁻⁵ (slightly soluble)
SrSO₄ – 3.4 × 10⁻⁷ (slightly soluble)
Carbonates: CaCO₃ – 3.4 × 10⁻⁹ (insoluble)
BaCO₃ – 2.6 × 10⁻⁹ (insoluble)
PbCO₃ – 1.5 × 10⁻¹³ (very insoluble)
MgCO₃ – 6.8 × 10⁻⁶ (slightly soluble)
Hydroxides: Fe(OH)₃ – 2.8 × 10⁻³⁹ (extremely insoluble)
Al(OH)₃ – 1.3 × 10⁻³³ (extremely insoluble)
Cu(OH)₂ — 2.2 × 10⁻²⁰ (very insoluble)
Zn(OH)₂ — 3.0 × 10⁻¹⁷ (very insoluble)
Mg(OH)₂ — 5.6 × 10⁻¹² (insoluble)
Ca(OH)₂ — 5.0 × 10⁻⁶ (slightly soluble)
Ba(OH)₂ — 5.0 × 10⁻³ (moderately soluble)
Phosphates: Ca₃(PO₄)₂ — 2.1 × 10⁻³³ (extremely insoluble)
Ag₃PO₄ — 8.9 × 10⁻¹⁷ (very insoluble)
FePO₄ — 9.9 × 10⁻¹⁶ (very insoluble)
Sulfides: CuS — 6.3 × 10⁻³⁶ (extremely insoluble)
PbS — 9.0 × 10⁻²⁹ (extremely insoluble)
ZnS — 2.9 × 10⁻²⁵ (very insoluble)
FeS — 6.3 × 10⁻¹⁸ (very insoluble)
MnS — 2.5 × 10⁻¹³ (insoluble)
EFFECT OF pH ON SOLUBILITY Rules
The pH value has a substantial impact on the solubility of substances containing ions reacting with either H⁺ or OH⁻. It is particularly noteworthy in the case of hydroxides, carbonates, phosphates, and sulfides.
How Acidic Conditions Increase Solubility
In a solution that contains an insoluble substance, adding an acid will cause the hydrogen ions (H⁺) to react with the anion of the substance.This removes the anion from the equilibrium, shifting the dissolution equilibrium to the right and dissolving more of the solid.
Example – Mg(OH)₂ in acidic solution:
Mg(OH)₂ (s) ⇌ Mg²⁺ (aq) + 2OH⁻ (aq)
Adding H⁺: H⁺ (aq) + OH⁻ (aq) → H₂O (l)
The H⁺ ions react with the OH⁻ ions and remove them from the solution. This causes the equilibrium to shift to the right, thus increasing the dissolution of Mg(OH)₂. This explains why insoluble metal hydroxides dissolve fully in acidic solutions.
Example – CaCO₃ in acidic solution:
CaCO₃ (s) + 2HCl (aq) → CaCl₂ (aq) + H₂O (l) + CO₂ (g)
That is precisely what happens when acidic rain reacts with limestone buildings and marble sculptures; acidic water dissolves insoluble calcium carbonate.
How Basic Conditions Affect Solubility:
In the case of substances such as metal hydroxides, an increase in pH, which means higher basicity, reduces their solubility. The addition of additional hydroxyl ions in a solution that already contains metal hydroxides shifts the equilibrium towards the formation of solids.
Example: Adding NaOH to a solution already saturated with Mg(OH)₂ adds more OH⁻ ions. This pushes the equilibrium back:
Mg²⁺ (aq) + 2OH⁻ (aq) → Mg(OH)₂ (s) ↓
More precipitate forms and solubility decreases.
Amphoteric Hydroxides – Special pH Behavior
A certain class of metal hydroxide salts dissolves not only in acidic but also in strongly basic environments. At neutral pH, they are insoluble but become soluble under very acidic or very basic pH values.
Examples of amphoteric hydroxides: Aluminum hydroxide Al(OH)₃
Zinc hydroxide Zn(OH)₂
Lead hydroxide Pb(OH)₂
Chromium hydroxide Cr(OH)₃
Aluminum hydroxide behavior:
In acid: Al(OH)₃ (s) + 3H⁺ (aq) → Al³⁺ (aq) + 3H₂O (l) – dissolves
In neutral: Al(OH)₃ – insoluble precipitate
In strong base: Al(OH)₃ (s) + OH⁻ (aq) → [Al(OH)₄]⁻ (aq) – dissolves as aluminate ion
EFFECT OF TEMPERATURE ON SOLUBILITY
There is a significant impact of temperature on solubility and can be explained through thermodynamics.
Solubility of Solids Increases With Temperature (Usually)
The dissolution of ionic compounds in aqueous solution is usually an endothermic process, one that requires heat. This means that, according to Le Chatelier’s principle, an increase in temperature will result in more product formation.
Examples of solubility increasing with temperature:
KNO₃ — 13 g/100 mL at 0°C → 247 g/100 mL at 100°C (dramatic increase)
NH₄Cl — 29 g/100 mL at 0°C → 77 g/100 mL at 100°C
KCl — 28 g/100 mL at 0°C → 57 g/100 mL at 100°C
NaNO₃ — 74 g/100 mL at 0°C → 180 g/100 mL at 100°C
Exceptions – Solubility Decreases With Temperature
There are few exceptions where some substances have an exothermic dissolution reaction, resulting in reduced solubility with rising temperatures.
Examples:
Na₂SO₄ – solubility increases up to 32°C then sharply decreases
Ce₂(SO₄)₃ – cerium sulfate becomes less soluble at higher temperatures
Li₂SO₄ – lithium sulfate shows slight decrease in solubility with temperature
Ca(OH)₂ — calcium hydroxide becomes less soluble as temperature increases
Solubility of Gases Decreases With Temperature
In case of gases dissolved in water, there will be a decrease in solubility as temperature increases. This is due to the fact that gas dissolution is always an exothermic process; energy is given out during the process of dissolving gas.
Examples:
O₂ in water – 14.6 mg/L at 0°C → 7.6 mg/L at 30°C (solubility nearly halved)
CO₂ in water – much more soluble at 0°C than at 40°C
N₂ in water – solubility decreases steadily with increasing temperature
EFFECT OF PRESSURE ON SOLUBILITY
Henry’s Law — Gas Solubility and Pressure
In the case of gases in a solvent, the solubility of the gas increases with an increase in the pressure of the gas. This principle is captured by Henry’s Law:
C = k × P
Where:
C = concentration of dissolved gas (mol/L)
k = Henry’s Law constant (specific to each gas and temperature)
P = partial pressure of the gas above the solution (atm)
Practical Examples:
Carbonated beverages are stored under high CO₂ pressure (3-4 atm). Upon opening the bottle, the pressure is lowered to 1 atm, resulting in the rapid escape of CO₂ from solution because of its decreased solubility due to the lower pressure.
In scuba diving, air is inhaled under higher pressure. Nitrogen gas enters the blood of the diver in direct proportion to the increased pressure. If the scuba diver rises to the surface too quickly, pressure is decreased rapidly, leading to the formation of nitrogen bubbles in the blood vessels, thus producing decompression sickness.
Pressure Has Little Effect on Solid Solubility
The solubility of solids and liquids in water is not dependent on the change in pressure in the atmosphere because unlike gases, the amount of volume change in the case of dissolving solids is very insignificant.
Solubility Rules Chart
The solubility rules chart can be considered one of the most useful reference tools for chemists. This table enables the user to predict immediately if any compound is soluble or forms a precipitate when dissolved in water. One does not need to remember how each and every single substance behaves; he or she just needs to know the type of ions.
How to Read the Chart?
The chart is divided into two main columns – the ion type and its solubility behavior – followed by any notable exceptions. To use it correctly, always identify the cation and anion of your compound first, then check the chart starting from the “always soluble” rules and working downward. If the cation appears in the always-soluble category, the compound is soluble regardless of the anion. If not, check the anion rules next.
Solubility rules in water
For aqueous solutions at room temperature and standard conditions
Soluble (>0.1 mol/L)
Slightly soluble / sparingly
Insoluble (<0.01 mol/L)

