What is Molality?
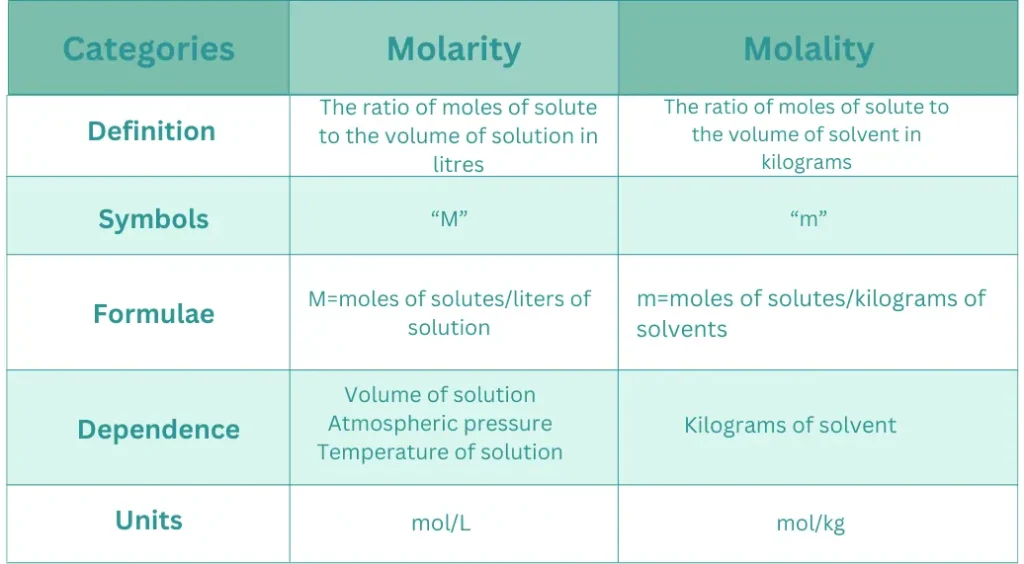
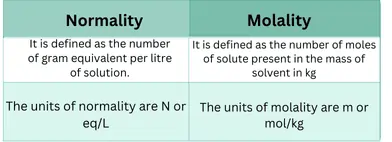
The molality of a solution is the number of moles of solutes per kilogram of solvent.
Molality Formula
Its formula is given below:
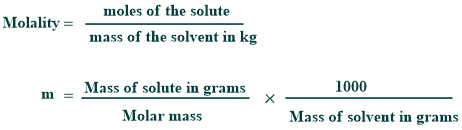
How to find molality
It can be calculated by using in three steps:
Step 1: Find the Number of Moles
With the use of given formula:

Step 2: Convert Solvent Mass into Kilograms
If the solvent mass is given in grams, divide it by 1000.
Step 3: Apply the Molality Formula
After the conversion of solvent mass into kilograms, apply the given formula:
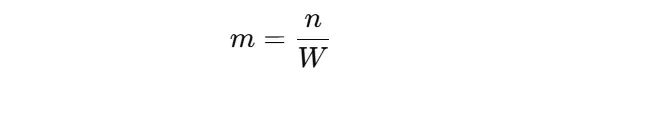
For Example,
Molality of pure water
As it is known, the density of pure water is 1 kg per litre, and its molar mass is 18 g per mole. Therefore,

However, pure water is 55.55 molal.
If a solution containing 5.3 grams of anhydrous Na₂CO₃ in 200 g of water, then its molality can be calculated as follows:

As a result, the anhydrous sodium carbonate solution is 0.25 molal.
Molality of Toluene Solution
Calculate the molality of a solution where 0.5 grams of toluene (C7H8) is dissolved in 225 grams of Benzene (C6H6). Calculate the moles of the given solute.
Solution:
Toluene: Molecular weight =
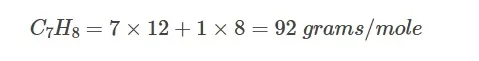
With the help of the given formula
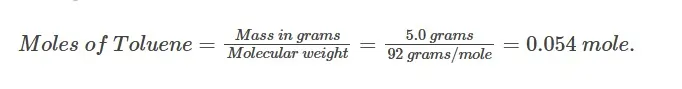
Therefore, the moles of toluene are 0.054.
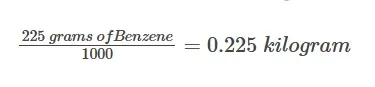
The final step is to calculate the molality of the toluene solution.

Applications of Molality
Molality is used in many chemistry and industrial fields.
Physical Chemistry
Scientists use molality for accurate concentration calculations.
Freezing Point Depression
It helps determine how much a solute lowers the freezing point.
Boiling Point Elevation
It is used to study boiling point changes in solutions.
Pharmaceutical Industry
Drug formulations sometimes use molality-based calculations.
Chemical Research
Researchers prefer molality in temperature-sensitive experiments.
Industrial Chemistry
Industries use molality for concentrated chemical mixtures.
What are the advantages and disadvantages of using molality?
Advantage
Disadvantage
Relationship between molality and molarity
Their relationship is given below:
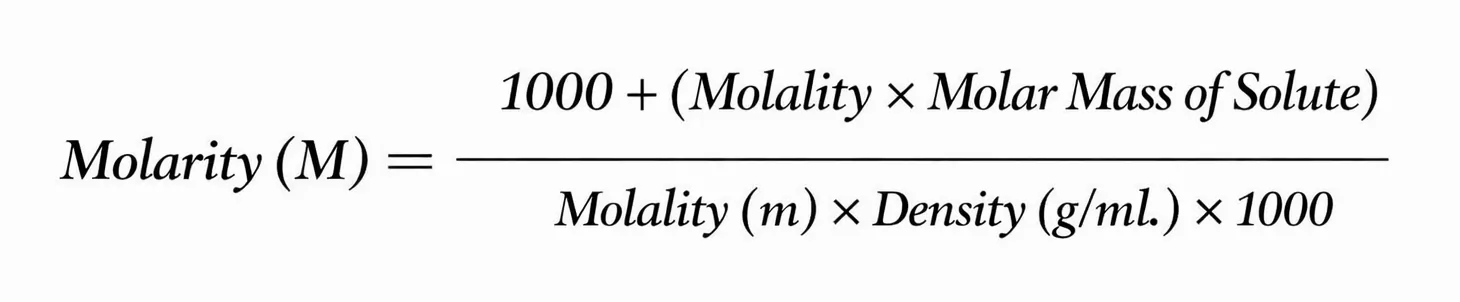
Where,
d = the density (g/ml)
m₁ = the molar mass of the solute
M=molarity
m=molality
Real-Life Example of Molality
Saltwater is a common example.
If you dissolve salt in water, the molality tells you how many moles of salt are present in one kilogram of water.
This concept is useful in:
Common Mistakes While Calculating Molality
Mostly, students make these mistakes:
Therefore, you should always double-check units before solving problems.