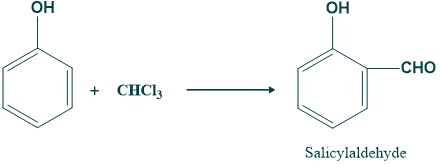
Riemann Reaction Mechanism:
Riemer reaction mechanism is nine step mechanism which are as followa;
Step(01):
In this step base (OH) attacks on chloroform and deprotonated chloroform
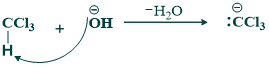
step(02):
In this step deprotonated chloroform remove chloride ion and give dichlorocarbene.
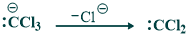
Step(03):
In this step dichlorocarbene act as electrophile. Phenoxide ion act as nucleophile and attack on dichlorobenzene.
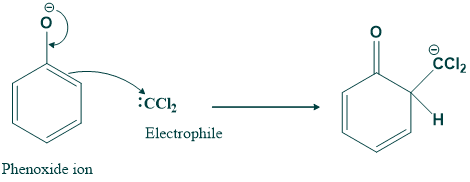
Step(04):
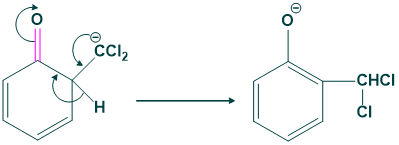
In this step shifting of hydrogen takes place
step(05):
In this step oxygen share its lone pair to carbon by removing a chloride ion.
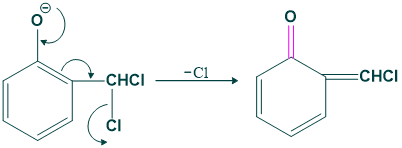
Step 06
In this step base attack on the product which are produced in step 05.
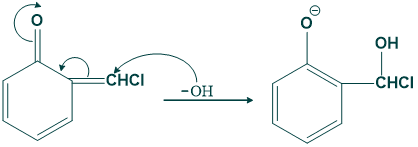
Step(07):
In this step oxygen give its lone pair to carbon by removing chloride ion as follows;
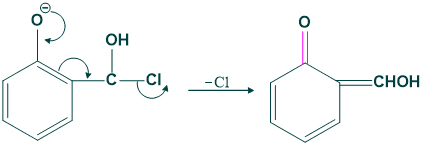
Step(08):
In this step attack of base takes place again.
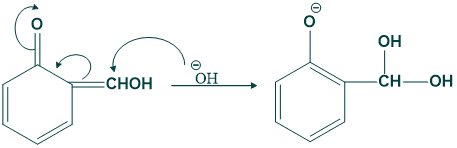
Step (09):
In this step removal of water takes place and give o-hydroxy benzaldehyde or salicylaldehyde.
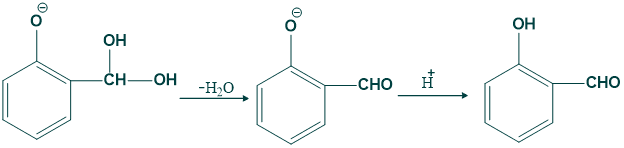

Riemann Reaction Mechanism:
Riemer reaction mechanism is nine step mechanism which are as followa;
Step(01):
In this step base (OH) attacks on chloroform and deprotonated chloroform
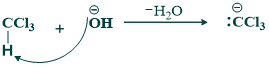
step(02):
In this step deprotonated chloroform remove chloride ion and give dichlorocarbene.
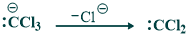
Step(03):
In this step dichlorocarbene act as electrophile. Phenoxide ion act as nucleophile and attack on dichlorobenzene.
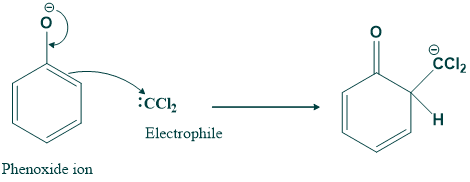
Step(04):
In this step shifting of hydrogen takes place

step(05):
In this step oxygen share its lone pair to carbon by removing a chloride ion.
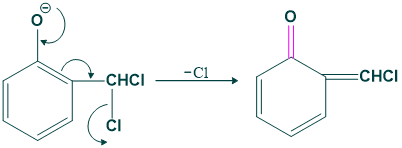
Step 06
In this step base attack on the product which are produced in step 05.

Step(07):
In this step oxygen give its lone pair to carbon by removing chloride ion as follows;

Step(08):
In this step attack of base takes place again.

Step (09):
In this step removal of water takes place and give o-hydroxy benzaldehyde or salicylaldehyde.