What is tautomerism?
- Tautomerism is a phenomenon in which a hydrogen atom’s movement and a double bond’s shifting take place.
- It refers to isomers that can easily interconvert.
- A chemical phenomenon in which a single chemical molecule exists in two or more interconvertible forms with different placements of a proton and double bond. These various structures are known as tautomers, and such processes are known as tautomerism in chemistry. Interconversion between these structures is often quick and occurs in a dynamic equilibrium condition.
- The most prevalent type of tautomerism is keto-enol tautomerism, which occurs when a molecule with a keto group (C=O) is transformed into its enol counterpart (C=C-OH). For example, in acetoacetic acid, the keto form is CH₃-CO-CH₂-COOH, while the enol form is CH₃-C(OH)=CH-COOH.
- It is a structural isomerism significant in many chemical and biological processes.
- “Tautomers” and “contributing structures in chemical resonance” are two different terms; don’t confuse them.
Examples of Tautomerism:
Keto-Enol Form:

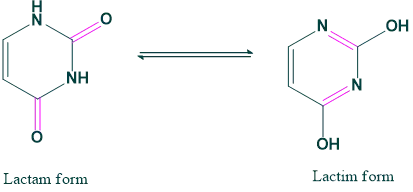
Lactam-Lactim form:
Amide-imidic acid form:

Amine-Imine form:

Conditions for tautomerism:
- That compound must contain an electron-withdrawing group that can abstract the hydrogen atom.
- The carbonyl group must contain an alpha hydrogen atom.
- The keto-enol tautomerism type proceeds with the availability of an acidic or basic catalyst.
Structural requirement of tautomerism in chemistry:
- Those compounds contain polar molecules and weakly acidic functional groups.
- Involvement of change in the position of an atom.
- It does not affect the bond length or such features.
- Normally, it occurs in planar or non-planar molecules.
Types of tautomerism:
1. Prototrophy:
The process that arises due to the transfer of a proton within a molecule, i.e., an amino acid, which is the building block of protein, is called prototrophy.

2. Annular tautomerism:
The process that arises due to the shifting in a heterocyclic system.
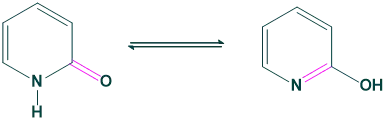
3. Ring chain tautomerism:
The phenomenon that arises due to the shifting of protons from the open chain and converting them into a closed chain.
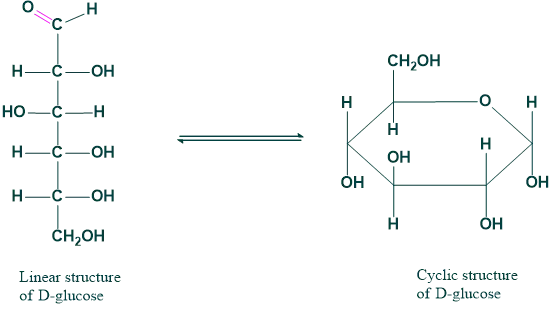
4. Non-carbonyl tautomerism:
The phenomenon that arises due to the shifting of protons in noncarbonyl compounds.



5. Valence tautomerism:
That phenomenon arises due to the continuous formation and breaking of single and double bonds in the compound without any migration of groups or atoms.
The molecular geometry of a molecule is changed in valence tautomerism and is not confused with canonical resonance structures.
6. Tautomeric form of unsymmetrical ketones:
There is only one form of tautomer that occurs in symmetric form. However, for an unsymmetrical form, there can be two tautomeric forms.
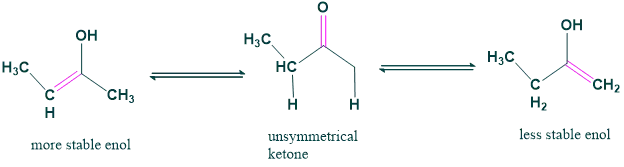
7. Keto-enol tautomerism:
- The process that arises due to the conversion of keto form into enol form by the action of a small amount of acid or base as a catalyst is called keto-enol tautomerism.
- Tautomers are constitutional isomers that are readily interconvertible. These are differentiated based on the placement of a labile hydrogen atom and the position of the double bond.
- The equilibrium between tautomers is not only rapid under normal conditions but also often strongly favors one of the isomers. For example, acetone is 99.999% keto tautomer and ~0.001% enol tautomer.

- The minor tautomer and the major tautomer are formed due to the chemical behavior of the compound.
- Traces of acids or bases are used as catalysts for the tautomeric equilibrium.
- A keto tautomer favors the rapid equilibrium more than the enol tautomer. But in some other reactions, enol is the most important tautomer.
Case 1: Intramolecular H-bonding
One example is the 1,3-diketones, such as acetylacetone (2,4-pentanedione), which prefer the enolic form. Intramolecular H-bonding creates a 6-membered cyclic structure that stabilizes the enol form over the keto.

Case 2: Aromaticity
In some aromatic compounds, i.e., phenol, the enol exhibits the aromatic character, so it is more favorable than the ketone.

Case 3: Solvent
Polar solvents (which can form hydrogen bonding) will make lone pairs less available for intramolecular H-bonding; hence, the percentage of keto increases. aromatic character

keto: enol
6: 94 (when benzene is used as solvent)
81:19 (when water is used as solvent)
Case 4: Conjugation
A conjugated enol is more favored for a ketone than a non-conjugated enol.
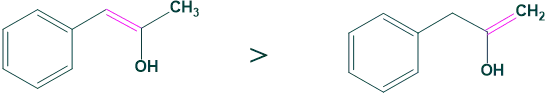
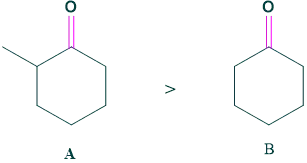
Case 5: Substitution
The more substituted ketone (A) is more favored for enol than (B) ketone.
Recommended videos:
part -1 :
Part 2