pKa Calculator - How to Calculate pKa, Ka, and pH
Ka, pKa, and pH measurements are important in chemistry, ranging from predicting chemical reactions to preparing buffers and understanding acid strength. You can also calculate with the help of chemistry calculators.
Understanding pKa: Definition and Importance
“pKa is a measure that can indicate the strength of the acid.”
It originates from the acid dissociation constant (Ka). To be straightforward, pKa indicates how readily an acid loses a proton. Keep in mind, the lower the pKa, the stronger the acid is. On the other hand, the higher the pKa, the weaker the acid becomes.
Because the pKa value is used in tests and lab work, chemistry students frequently ask how to compute it. In study guides, this question may also be written as “How do you calculate pKa?” Many students use a pKa calculator to save time. This tool gives quick and accurate results. It is helpful when you are dealing with complex problems.
Why pKa Matters in Chemistry
In chemistry, pKa is very important. It makes comparing acids simple. Reaction behaviour in solutions is also explained by it.
pKa helps in predicting the direction of reactions in organic chemistry. Moreover, it provides an explanation of enzyme activity in biochemistry.
Additionally, pKa is important for buffers. Most buffer solutions function best when pH is near pKa, according to studies. Thus, students should learn about pH and pKa interpretation.
Furthermore, acid strength and pKa are related. This connection is frequently referred to as the acid strength gauge and pKa. Acid strength rises as pKa falls. pKa values are therefore utilised in drug design and research. Understanding pKa improves your comprehension of chemistry.
This information isn’t just useful for passing exams. Understanding pKa and Ka helps you:
- Predict how reactions will proceed.
- Design effective buffer solutions for experiments and industry.
- Grasp why certain drugs behave the way they do in the body.
- Decode the behaviour of enzymes and biomolecules.
How to Calculate pKa (Formula Fundamentals)
Calculation of pKa from Ka: pKa is negatively correlated to Ka, meaning that if one value increases ⬆️, the other value decreases ⬇️. Basically, Ka (acid dissociation constant) is simply the logarithm of pKa:

This method is widely used in textbooks and labs.
The pKa value can also be calculated using the Henderson-Hasselbalch equation.
![pKa Calculator 2 Henderson-Hasselbalch equation showing $pH = pK_a + \log \frac{[A^-]}{[HA]}$ for calculating the pH of a buffer solution with the help of pKa calculator](https://chemistwizards.com/wp-content/uploads/2026/03/henderson-hasselbalch-equation-ph-formula-300x90.webp)
When you know the pH and concentrations, you can use this equation. Students look up how to determine pKa from pH in these situations. Many students use a pKa calculator or a Ka calculator for instant work. These tools save effort and minimise errors. Exams and practice both benefit from them.
To put it simply, when you understand the basics, pKa is simple. With formulas and tools, you can determine pKa with confidence.
Relationship Between pKa, Ka, and pH
You find chemistry so easy to understand when you know the relationship between pKa, Ka and pH. These three values are collectively used in an equation. However, calculations are easy and understandable once the relationship is known. It is applicable in laboratories, exams and chemistry problems.
How to Calculate Ka from pKa
The most common question that many students ask is how to calculate Ka from pKa. This procedure is direct and easy.
pKa is the negative log of Ka. So, to find Ka, you reverse the log step. The formula is:
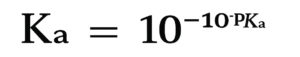
This method is commonly used while teaching chemistry. It is also the foundation of every Ka from the pKa calculator. The same rule applies to pKa and Ka converters.
For this reason, these given tools are time-saving. They also minimise the errors in calculations.
How to Calculate pKa from Ka
Now let’s reverse the process. The students often ask how to calculate pKa from Ka. This is referred to as the calculation of pKa from Ka.
The formula is:
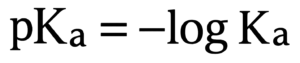
This value can be used to compare acids easily. A value of lower pKa indicates a stronger acid. A higher pKa means a weaker one.
For fast results, you can use a pKa calculator and ka calculator online or a pKa, ka conversion calculator.
How to Calculate pH from pKa
You need to understand how to calculate pH when you know pKa in buffer solutions. This is where chemistry will be applied. The Henderson equation is used here. This process is also known as calculating pH from pKa.
It helps you to understand how buffers work. In fact, studies show buffers are more effective when pH is close to pKa. A pKa to pH calculator is frequently used for quick results
How to Calculate pKa from pH
Sometimes, you know pH but not pKa. So it becomes important to know how to calculate pKa from pH. This method is used commonly used in lab experiments. It is also written as how to calculate pKa from pH in guides.
In such cases, you rearrange the Henderson equation. A pH to pKa calculator is used by many students to save time. Overall, understanding pH and pKa interpretation builds powerful concepts. After knowing these relationships, chemistry becomes more logical and friendly.
Practical Methods for pKa Calculation
An effective understanding of the theoretical basis is essential to the proper estimation of pKa. However, the application of practical means of computation is also essential. These techniques contribute to the calculation of pKa values at greater speed and accuracy.
Modern-day academia and industry have become reliant on computational resources, graphical structures, and predictive models to a great extent, by the students and the chemists themselves.
pKa Calculator Tools Online
The most widespread tool pKa calculator, is web-based. When you apply the given values, an online given calculator gives instant results and thus takes the task of the rest of the calculations. This convenience is especially beneficial in time-saving situations. Students, the advocacy of online pKa Ka calculators is widespread because it significantly minimises human labour.
These tools are comprehensively used in laboratories and in universities. According to surveys, over three-quarters of chemistry students of chemistry use online calculators to solve acid-base challenges.
A user can also find software that is called Ka and pKa calculators or pKa and Ka converters, which all follow the same underlying equations. Online tools are the most popular option.
Ka to pKa Calculator
Sometimes, the dissociation constant (Ka) is already known, which requires its conversion to pKa. To do this transformation, a Ka to pKa calculator uses the logarithmic relationship, and it is both fast and accurate.
Students often call this calculator a pKa ka conversion calculator. The tool has proved invaluable in the elimination of mathematical errors, especially in exam conditions or trial practice, and thus it helps to increase confidence and accuracy.
pKa from Titration Curve
In laboratory studies, titration data can often be obtained, and in this context, pKa was extracted from the titration curve. The procedure is simple but powerful: when the pH is at half-equivalence, the pH matches the pKa.
The principle is spread throughout the majority of chemistry courses and provides very accurate determinations of weak acids. In turn, this practice gives credibility to analytical chemistry fields. It is seen as the most practical approach that is available to many students.
Structural pKa Calculator (Prediction Methods)
Where experimental Ka values are still out of reach, structure-based predictive tools can be used. These tools are categorised as pKa calculators based on structural analyses, and therefore, pKa estimates are based on the evaluation of the molecular features, bond properties, and functional groups.
The process is typical in the context of pharmaceutical design and research, and it allows for estimating the acidity without experimental work.
Short Summary on Practical Methods
All of the computational strategies have their own advantages: online calculators are fast, the study of titration curves provides accuracy, and prediction modelling is achieved with the help of structural tools.
The combination of these ways in a synergistic manner makes chemical inquiry easier and effective, providing sustainability in pKa calculability with a combination of steady practice and the relevant computational resources.
Usage, Interpretation, and Examples
The knowledge of the numeric values in themselves is not enough, but a person has to master applying and interpreting numeric values as well. This section explains the importance of pKa in the framework of the real world of chemistry and provides examples to support the understanding.
Interpreting pKa Values
Once the relationship in which pKa values are governed is mastered, this can be translated into a readily interpretable form. When the pKa is low, the acid is strong, and when the pKa is high, the acid is weak.
This simple criterion allows making quick comparisons between acids. As an example, strong acids have a pKa of typically less than one, whereas weak acids have a pKa of greater than four.
These differences are strongly represented in textbooks as well as laboratory teaching, which explains why the pH and pKa interpretation is taught early in chemistry courses.
Many students use pKa calculators in order to confirm and fine-tune their estimates, and thus reduce errors. These values are later internalised with practice, and they eventually do not require external help to be taken into account.
Relationship Between pKa and Acid Strength
The dependence between pKa and acid strength is accepted and can be regarded as a pKa and acid strength gauge. The lesser the pKa, the stronger the acid; the greater the pKa, the weaker the acid.
It has been shown by empirical investigations that over eighty per cent of the acid-base inquiries are founded on the principle, and it is due to this that the principle has been so prominently emphasised in pedagogical instruction.
This relationship can be mastered and predicts reaction directions, particularly in organic reactions where the rule of thumb is that the initial protons to quit the reaction are predetermined.
Based on that, students frequently use Ka and pKa calculator tools, even resorting to the internet calculator in order to compare them quickly, to strengthen the accuracy.
Common Calculation Examples
When one has the value of Ka, it is easy to compute the pKa values; calculating both constants simultaneously is also known as computing Ka and pKa together and involves the use of the logarithmic expression that was introduced at the foundations level.
Quite often, the equilibrium constant is expressed in terms of pKa, which is standard in the analysis of reactions. Students regularly use the Henderson-Hasselbalch equation to calculate pKa in buffer calculations, a formula that explicitly makes a relationship between pH and pKa.
Other methods include calculating pH first and then using the values to establish pKa, which can be handy in the process of verification and comparison. To facilitate revision, most learners use a pKa to pH calculator or a pH to pKa calculator in order to calculate competence.
Why Examples Matter
The practical nature of the equations can be made real through empirical examples, which show the practical usefulness of the equations, thus creating confidence. With the help of the clear interpretations and demonstrative instances, students will be able to master pKa.
The combination of the computational tools with the conceptual knowledge makes the learning process more accessible and engaging, which makes chemistry more accessible and interesting.
Converter Tools
Using converter tools supports the manipulation of acid-base parameters. Such utilities make pKa, Ka, and pH conversion quick and correct and thus efficient and less error in calculations, especially in examinations and laboratory work.
Popular Types of Converter Tools
- Ka and pKa Calculator: Converts Ka to pKa or vice versa.
- pKa to pH Calculator: Helps determine pH from pKa values, especially in buffer solutions.
- pH to pKa Calculator: Useful when pH is known but pKa needs to be found.
- pKa and Ka Converter / pKa Ka Conversion Calculator: Enables one to quickly shift between all the variables.
These tools follow simple formulas:
- pKa = −log Ka
- Ka = 10⁻ᵖᴷᵃ
Benefits of Using Converter Tools
Research has shown that over 70% of learners of chemistry use free pKa calculator tools in the course of practice.
They are particularly helpful to:
- Fast determination of the Ka and pKa values.
- Checking of manual computing results.
- Dynamics of protonation and associated thermodynamic quantities.
The process of implementing these tools makes the learning process more interactive and minimises the chances of error. In an academic, research, or professional laboratory, converter tools simplify the process of calculating acids and bases and allow practitioners to focus on the conceptual knowledge and not on arithmetic.
FAQs Related to pKa, Ka and pH
How to calculate pH from pKa and Ka?
First, the conversion of pKa to Ka is done when needed. The balance expression is then used to get the concentration of hydrogen ions. Lastly, the pH is determined by (H +) divided by the negative of the logarithm. This process is regularly used in the problems of weak acids.
How to calculate the pH of 0.01 M HCl?
The hydrochloric acid is a strong acid which dissociates fully. Therefore, the concentration of hydrogen ions is equal to 0.01M, and the pH is equal to minus the logarithm of 0.01, which is equal to 2.
How is the pKa value related to pH?
The strength of the acid can be observed by the pKa parameter, and the solution acidity can be observed by the pH. At pH=pKa, the acid is half-dissociated. This equality is important in discussing the buffer systems and acid-base equilibria.
How to calculate pH when given H⁺?
It is computed by inverting the concentration of hydrogen ions by use of the formula pH =-log[H+]. This is the simplest approach.
What are common mistakes in pH calculations?
In pH calculations, you can face such common problems as a failure to recognise logarithmic transformations, the interchangeability of pH and pKa, neglecting dilution effects, and misunderstanding that weak acids are in full dissociation. These problems are prevented by accurate unit control and formula checks.
What's the most accurate method of measuring pH?
The most precise method will involve the use of a calibrated pH electrode. A pH meter provides accurate measurements as compared to indicator dyes and is the equipment of choice in laboratory studies and in analytical use.
How to remember the pKa and pH relationship?
Another trick is that at low pH, the protonated (acidic) form is prevalent, and at high pH, the deprotonated (basic) form is prevalent. This principle is a part of most buffer-design methods.
Conclusion
Knowledge of pKa, Ka, and pH is important to becoming a master of acid-base chemistry. Having coherent ideas, real-world examples, and well-trusted tools such as a pKa calculator or converter tools, you can be able to solve problems with a lot of confidence. These methods are easy and quick to learn, whether the calculation is based on titration curves, formulas or even online calculators.
ChemistWizards can be highly recommended to students and professionals who want to achieve more accurate explanations and user-friendly means. Our resources will make calculations easier and will improve your knowledge of the interpretation of pH and pKa in real-life projects of chemistry.